209 Polyphenol-metal Complex With Dopamine
time can still be achieved when the natural white hair time of ethanolamine soaking is 30
minutes, and the dyeing time of GA-Fe(II)+DA hair dye is 30 minutes. The dyeing time
can be adjusted according to the dyeing needs in the actual application process.
HAIR DYEING MECHANISM AND ANALYTICAL CHARACTERIZATION
To gain a deeper understanding of the self-assembly process involving GA and ferrous
chloride tetrahydrate, which undergo a coordination reaction in solution to form a
polyphenol-metal complex, and to confirm that dopamine remains independent in the
solution without participating in the coordination reaction, we prepared GA-Fe(II)+DA
solutions and analyzed their UV-Vis absorption spectra. This analysis aimed to illustrate
that GA-Fe(II) successfully reacts to complete the self-assembly process, while dopamine
does not.
The UV-Vis absorption spectrum of GA-Fe(II)+DA solution has a broad absorption
ranging from 800 to 500 nm, along with a coordination-to-metal charge transfer (LMCT)
band at 621 nm. These findings indicate the in-situ formation of GA-Fe(II) coordination
structure and the predominant presence of bi-type coordination within the polyphenol-
metal complex (Figure 6C). The shoulder peak at 230 nm–290 nm demonstrated the
intact presence of dopamine. The composite solution of GA-Fe(II)+DA and GA-Fe(II)
solution (without DA addition) were centrifuged (2,000g, 5 min) and the supernatant
was freeze-dried (SCIENTZ, SCIENTZ-12N, Freeze-dryer) to obtain FR-IT spectra for
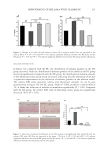
Figure 4. (A), (B), (C) ethanolamine optimal pH screening. (D) Screening for optimal concentration of
ethanolamine. (E) The time for which ethanolamine soaks natural gray hair.
time can still be achieved when the natural white hair time of ethanolamine soaking is 30
minutes, and the dyeing time of GA-Fe(II)+DA hair dye is 30 minutes. The dyeing time
can be adjusted according to the dyeing needs in the actual application process.
HAIR DYEING MECHANISM AND ANALYTICAL CHARACTERIZATION
To gain a deeper understanding of the self-assembly process involving GA and ferrous
chloride tetrahydrate, which undergo a coordination reaction in solution to form a
polyphenol-metal complex, and to confirm that dopamine remains independent in the
solution without participating in the coordination reaction, we prepared GA-Fe(II)+DA
solutions and analyzed their UV-Vis absorption spectra. This analysis aimed to illustrate
that GA-Fe(II) successfully reacts to complete the self-assembly process, while dopamine
does not.
The UV-Vis absorption spectrum of GA-Fe(II)+DA solution has a broad absorption
ranging from 800 to 500 nm, along with a coordination-to-metal charge transfer (LMCT)
band at 621 nm. These findings indicate the in-situ formation of GA-Fe(II) coordination
structure and the predominant presence of bi-type coordination within the polyphenol-
metal complex (Figure 6C). The shoulder peak at 230 nm–290 nm demonstrated the
intact presence of dopamine. The composite solution of GA-Fe(II)+DA and GA-Fe(II)
solution (without DA addition) were centrifuged (2,000g, 5 min) and the supernatant
was freeze-dried (SCIENTZ, SCIENTZ-12N, Freeze-dryer) to obtain FR-IT spectra for
Figure 4. (A), (B), (C) ethanolamine optimal pH screening. (D) Screening for optimal concentration of
ethanolamine. (E) The time for which ethanolamine soaks natural gray hair.