211 Polyphenol-metal Complex With Dopamine
for the complex molecules to penetrate the hair cortex layer (Figure 7B). By unfolding
the hair scales using ethanolamine emulsion, the maximum critical molecular size that
the protective layer of hair scales can allow to pass through increases, and large-size dye
molecules can also easily penetrate the hair cortex layer to achieve natural white hair
dyeing. Dopamine, which is free in the hair dye, also penetrates the cortical layer of hair
and is deposited and autoxidized to polymeric polydopamine, which increases dramatically
in size and cannot penetrate from the cortical layer to the hair surface through the hair
scales. Its adhesive properties effectively secure the dye molecules within the cortical layer
of the hair, accomplishing color fixation, enhancing the hair’s resistance to washing, and
resulting in a permanent dyeing effect.
In our study, we observed that natural white hair dyed solely with GA complexed with
Fe(II) dye molecules exhibited poor resistance to washing, with a significant color difference
occurring even after just 40 washes. Dopamine can adhere to the surface of the hair and
form an adhesive film that acts as a color fixation.31 Therefore, we introduced dopamine
to prepare a composite hair dye for improving the problem of poor washing resistance and
achieving permanent hair dyeing. However, in our study, we found that dopamine did not
adhere to the hair surface, but rather entered the hair cortical layer through the hair scales.
Based on the property that dopamine is prone to spontaneous oxidation and polymerization
into polydopamine,32 we hypothesized that dopamine accumulates in the cortical layer
and undergoes autoxidation and polymerization into polydopamine, which adheres to fix
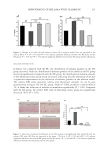
Figure 6. UV-Vis absorption spectra of (A) DA, (B) GA, and (C) GA-Fe(II)+DA. (D) FT-IR spectra of DA,
GA, Fe(II), GA-Fe(II)+DA.
for the complex molecules to penetrate the hair cortex layer (Figure 7B). By unfolding
the hair scales using ethanolamine emulsion, the maximum critical molecular size that
the protective layer of hair scales can allow to pass through increases, and large-size dye
molecules can also easily penetrate the hair cortex layer to achieve natural white hair
dyeing. Dopamine, which is free in the hair dye, also penetrates the cortical layer of hair
and is deposited and autoxidized to polymeric polydopamine, which increases dramatically
in size and cannot penetrate from the cortical layer to the hair surface through the hair
scales. Its adhesive properties effectively secure the dye molecules within the cortical layer
of the hair, accomplishing color fixation, enhancing the hair’s resistance to washing, and
resulting in a permanent dyeing effect.
In our study, we observed that natural white hair dyed solely with GA complexed with
Fe(II) dye molecules exhibited poor resistance to washing, with a significant color difference
occurring even after just 40 washes. Dopamine can adhere to the surface of the hair and
form an adhesive film that acts as a color fixation.31 Therefore, we introduced dopamine
to prepare a composite hair dye for improving the problem of poor washing resistance and
achieving permanent hair dyeing. However, in our study, we found that dopamine did not
adhere to the hair surface, but rather entered the hair cortical layer through the hair scales.
Based on the property that dopamine is prone to spontaneous oxidation and polymerization
into polydopamine,32 we hypothesized that dopamine accumulates in the cortical layer
and undergoes autoxidation and polymerization into polydopamine, which adheres to fix
Figure 6. UV-Vis absorption spectra of (A) DA, (B) GA, and (C) GA-Fe(II)+DA. (D) FT-IR spectra of DA,
GA, Fe(II), GA-Fe(II)+DA.