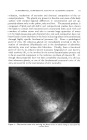
AMPHOTERIC SURFACTANTS: FATTY AMINO ACID DERIVATIVES 279 As is the case with all surface active agents, hydrophilic-hydrophobic balance is an important factor in the electrochemical and performance characteristics of the fatty amino acids. The availability of a wide variety of fatty chains (obtained readily through fractionation of natural products) and of monoadduct and diadduct forms makes possible a very wide range of hydrophilic-hydrophobic balance. A given fatty chain can be balanced with an amine and one or two carboxylic groups. Also, blends are possible. PERFORMANCE CHARACTERISTICS Basically, surface active properties are derived more or less directly from the electrochemical nature of the surfactant. In the case of the fatty amino acid derivatives, this dependence is especially pronounced. Prop- erties based on fatty chain length and chemical structure (monoadduct or diadduct, type of salt) can be modified or even altered drastically with changes in pH. High-foaming products can be changed to low-foaming, good wetting to poor wetting, nonsubstantive to substantive, etc. ,4. Solubility. Commercially, these materials are available as sodium i o •0 o o o •-•x (c) = (D) 50- 0 ppm •ater 30øC. 40 X / /i/•'x \ /I I 30 (A) [ , xx x ,/•X---•- X /• •-• ß '½x" zo- (B) •,• •,//• I i I i 2 4 6 8 I0 12 pH = (E) Figure 1.---Solubility vs. pH of typical fatty amino acid derivatives. (A) Disodium N-lauryl g-iminodipropionate. (B) Sodium N-lauryl g-aminopropio- nate. (C) Disodium N-tallow/%iminodipropionate. (D) Sodium N-tallow aminopropionate. (E) Sodium N-hydrogenated tallow/%aminopropionate.
280 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS salts (flaked solids) or triethanolamine salts (aqueous solutions). The sodium salts are quite alkaline in reaction, much as are the fatty acid so- dium soaps. The triethanolamine salt solutions are highly self-buffered at a pH of about 8. The fatty amino acids, as the sodium salts, are gen- erally quite water soluble, with the dJadducts exhibiting significantly greater solubility than the monoadducts. In addition to the influence of the fatty chain and that of the monoadduct or diadduct, pH is a very impor- tant factor in determining solubility. Typical solubility characteristics are outlined in Fig. 1. B. Compatibility. By nature of their amphoterism, the fatty amino acid salts exhibit compatibility over wide ranges with most other surfac- rants. This compatibility is of special importance in the formulation of cosmetic products. The alkyl aminopropionates are compatible com- pletely with nonionic surfactants. They are compatible with soluble fatty acid soaps in all but the higher soap-to-amino-acid ratios. They are compatible with most anionic surfactants except in relatively narrow ranges at or near equal ratios of amino acid and anionic product. One of the outstanding compatibility characteristics of the amphoteric products de- scribed here is their compatibility with cationic surface active agents. The alkyl aminopropionates do not complex or precipitate cationics as do the anionic surfactants. This compatibility is not limited to physical form, but is shown in the fact that the substantive nature or germicidal effectiveness of the cationic is not interfered with even in formulations con- taining as much as a 15 to 1 ratio of fatty amino acid salt to cationic. Hence, it is possible to formulate products exhibiting specific cationic prop- erties containing quite low concentrations of cationic surfactant. C. Foam. Sodium lauryl •5-aminopropionate represents a very high foaming synthetic detergent ideally suited to the formulation of all types of shampoos. This product, either as its sodium or triethanolamine salt, exhibits a high degree of hard water stability (clear solutions and unim- paired foaming in 300 ppm. water). Foam volume is maintained over a wide range of concentration and pH. The corresponding diadduct, di- sodium lauryl •5-iminodipropionate, is an excellent foaming agent, although exhibiting a somewhat lower overall •)aming power than the monoadduct. The coco and tallow derivatives evidence proportionately less foaming power as might be expected from their longer fatty chains. D. Substantivity. Of very great interest in the realm of cosmetic chem- istry is the affinity of the fatty amino acid molecule for the skin or hair. This affinity is most likely attributable to the similarity in amphoterism of the epidermis and the fatty amino acid. This affinity is manifested in a series of interesting and useful properties. These products exhibit sub- stantivity to the hair, thereby imparting manageability or softness. By virtue of being substantive and because of their ability to exhibit cationic
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)