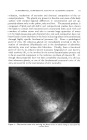
312 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS correlation has been obtained in comparing critical volume concentrations with calculated void spaces (4). Particles which are nearer to spherical shapes reach maximum dilatancy at higher solids concentrations cubes and more irregular shapes have lower concentrations for maximum effect, pre- sumably because they are able to pack down closely at rest and create larger voids when disturbed. Shearing stress Figure 5.--Model of dilatant flow, Dilatancy seems to require that the suspended particles be deflocculated the mechanism, as indicated in the "packing" idea discussed above, is en- tirely different from that assumed tk)r plastic or thixotropic flow. It is not surprising, therefore, that some pigment dispersions show both properties. Aqueous suspensions of carbon black, zinc oxide and iron oxide are known which give a dilatant curve with a loop. The types of flow we have just discussed in some detail are important be- cause we know that many problems of formulation and production involve the effect of small variables on the flow characteristics of industrial prod- ucts. We should next consider some of the common raw materials which are important in establishing or modifying the rheological pattern of our products. Gelling agents or thickening agents are an important group of modifiers. Concentration of the gelling agent may influence the nature of flow, as for example starch. Pastes containing 10 per cent dry starch form pseudo- plastic bodies and yet when the amount is increased up to 35 per cent or 40 per cent, they become entirely dilatant. Such pastes can be poured readily, but they seem to solidify if an attempt is made to stir them vigor- ously. Starch gels also vary somewhat according to the history used in
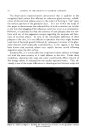
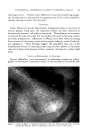
RHEOLOGICAL REVIEW FOR COSMETIC CHEMISTS 313 making thein up as shown in Fig. 6. Curve ,q is a 10% starch paste which was cooked for thirty minutes, while curve B is the same starch with a one hour cooking period. Dependence on method of preparation is common among the hydrophilic colloid gelling agents, and it points up the need for accurate theological knowledge as a control means for good quality produc- tion in industrial work. Ordinary gelatin also tends to change type of flow with concentration. A 4 per cent water dispersion of gelatin gives an excellent thixotropic curve, but higher concentrations approach dilatancy rather rapidly. Again there is a noticeable sensitivity in the type of gelatin chosen and the method used in gel formation with respect to flow properties. Aging of both starch and gelatin pastes leads to very definite changes in their theology. Dispersions of both sodium alginate and methyl cellulose show pseudo- plastic flow, somewhat similar to starch. Most water soluble gums are in the same theological family, though frequently they are mistakenly called thixotropic. Sheanng stres Figure &--Pseudoplastic flow of 10% starch pastes. Curve .4 = one hour cook. Curve B = half hour cook. Shearing stress Figure 7. 2.5% Ben-A-Gel in water. The mineral type of gelling agent usually behaves somewhat differently. Bentonite, or a refined magnesium montmorillonite, such as Ben-A-Gel, gives a definite thixotropic loop. Fig. 7 shows the flow curve of a suspen- sion of 2.5 per cent Ben-A-Gel in water. There is no evidence of any other type of flow at higher concentrations up to a virtually solid gel. Assuming the proper minimum work is done initially to forin the gel, no effect is no- ticed from variations in temperature or milling times on the theological properties. Stability on standing is a remarkable feature since duplicate measurements can be obtained after extremely long periods of time.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)