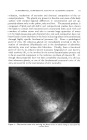
GAS-PARTITION CHROMATOGRAPHY. APPLICATIONS TO ESSENTIAL OILS AND OTHER VOLATILE MATERIALS* By C. L. TEITELBAUM Battelle Memorial [ns/i/u/e, Columbus Z, Ohio THE?,E HAVE BEEN a number of papers on the basic principles of gas- partition chromatography. The purpose of this paper is to discuss the application of. gas chromatography to analysis and research on essential oils and aromatic chemicals as well as other volatile materials. Figure 1 illustrates the basic principles and operation of a gas chromato- graph. It shows the essential parts: a cylinder of helium with a pressure regulator and flow meter sample introduction system, generally with flash heater and a column itself which is in many cases coiled or U-shaped, and is packed with a carefully chosen adsorbent material. Finally there is a detector cell, which is in two parts the reference cell through which the helium passes first, and the actual sensing part of the cell through which the product and the helium finally pass. Basically, there are two ways that the gas chromatograph can be used. It can be used primarily to obtain the chromatogram as a record for quali- tative and/or quantitative purposes. It can also serve as a means of ob- taining samples of various components of mixtures, with the chromatogram as a guide to the operation of the sample-collection system. Examples of chromatograms are discussed in conjunction with Figs. 2 and 3. A gas chromatogram of a sample of octyl aldehyde is shown in Fig. :2. This and the other chromatograms shown later have been changed so that they read from left to right, although, normally, recording machines will record from right to left. The first pip is the air peak then there are two very small peaks, and finally the very large octyl aldehyde peak. This, plus the shape of the major peak, is a good sign that we have quite pure octyl aidehyde. Since the octyl aldehyde peak is so smooth, any other purity would have to be extremely close to octyl aldehyde in retention time in order for its peak to be "hidden " the chances for this are very slim. Figure 3 shows an example of a good separation of a mixture of corn- * Presented at the December 13, 1956, Meeting, New York City. 316
GAS-PARTITION CHROMATOGRAPHY 317 PRESSURE REGULATOR TO SAMPLE COLLECTION SYSTEM I I.IELIUM CYLINDER THERMAL SAMPLE CONDUCTIVITY NTRODUCTION CELL REFERENCE SENSING CONSTANT TEMPERATURE BATH COLUMN Figure 1. pounds: methyl, ethyl and isopropyl benzoates. Their boiling points are respectively, 199 ø, 212 ø and 218øC., so that they cannot be separated nearly so cleanly by distillation. 'Fhis chromatogram was run at 190øC. with a column of crushed fire brick on which was suspended Apiezon wax. The chromatogram shown in Fig. 4 was rur• at a higher temperature, 228øC., on the same column. The ethyl and the isopropyl peaks are pushed together and the resolution is much poorer. Operation at a much lower tempera- ture, 167øC., is shown in Fig. 5. Here the peaks are spread out and are not nearly so amenable to quantitative interpretation in addition, the whole process is much slower. Figure 6 shows a chromatogram using a column material at 190øC. that did not give as good a separation. Of course, the peaks in the various chromatograms do not identify the particular compounds. They can be used for this purpose only if reference chromatograms of the individual compounds obtained under the identical conditions are available for comparison, and even then considerable caution must be exercised. If unknown compounds are involved, there is obvious need for complementary analysis of fractions corresponding to each peak, e.g., by infrared spectroscopy. In any case, the type of column packing and the operating condit}op.s must be chosen with care if best results are to be obtained.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)