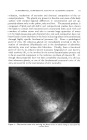
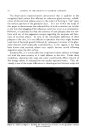
.324 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Figure 26.--"Tail end" of gas chroma- tography of Algerian (•/) and Reunion (B) geranium oils. Figure 27.--Gas chromatography of (a) pure lavender oil, (b) sophisticated lavender oil. ,outside the range of differences to be expected from one batch of pure oil to another. STUDY OF ESSENTIAL OIL COMPOSITION BY ISOLATION OF COMPOUNDS Another area of great potential utility for gas chromatography is in the investigation of the compositions of essential oils. Here it must be stressed that gas chromatography itself is not the complete solution to the problem of separating all the components of such complex mixtures. It is almost always necessary to use an approach based on a number of different separa- tion techniques applied in proper sequence. This is especially true in deal- ing with the minor components. For such purposes, gas chromatography can be regarded as a novel and extremely efficient distillation technique which routinely has a separation efficiency equivalent to 1000 to 2000 theoretical plates. Conventional distillation cannot begin to approach this efficiency. In addition, azeotropes do not form under the conditions of gas chromatography, and small amounts of material can be handled with ease. There are distinct differences in operation of a gas chromatograph when used as a separation tool, as compared with its use as an analytical or char- acterization tool. When used for analysis or characterization, it does not really matter what the chromatographic peaks represent reproducibility of the peaks is the main concern, whether they represent actual components of the original mixture or their decomposition products. However, if the aim is to identify the actual components, one must be certain that it is they that are being recovered, not their decomposition products. There are several ways by which one can detect a decomposition problem in such work (frequently caused by too high a column temperature). One
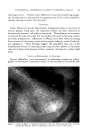
GAS-PARTITION CHROMATOGRAPHY 325 ran sometimes tell from the odor of a recovered fraction whether it is a de- tomposition product. If the fraction contains an olefin, occurrence of de- [tydration of an alcohol during the chromatography may be indicated. In- frared spectroscopy can be very helpful in this respect. For example, if two or three absorption peaks are present in the infrared spectrum of the •solate that are also present in the spectrum of the original mixture, it is at good indication that the isolate was present as such in the original mixture. Questions about differences in the structure and/or composition of rho- 12inol and citronellol have been puzzling essential oil and terpene chemists literally for decades. The two materials have markedly different odors, •vhich is the reason rhodinol costs eight or ten times as much as citronellol. Some investigators have ascribed the difference in odor solely to an isomeric Idifference between the major components citronellol possesses the isopro- Ipenyl structure, while the major component of rhodinol has been thought !by some to be an isomer of citronellol with an isopropylidene structure. lOther investigators have maintained that the difference in odor is due pri- •marily to desirable "impurities" or minor components of rhodinol that are mot present in citronellol. Some earlier work (without benefit of gas Ichromatography) had led us to agree with the latter explanation, but un- •equivocal evidence was still lacking. Figure 28 shows a chromatogram of one of the rhodinol samples obtained [for this work. This sample was unusual in that it did not give a sharp [peak but a rounded one as well as some unexpected peaks earlier in the [Figure 28.--Isolative gas chromatography of "abnormal" rhodinol. Figure 29.--Isolative gas chromatography of rhodinol I
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)