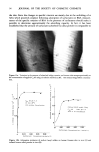
62 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS ENZYME INHIBITION We have reported (15) an inhibitory effect of various common anionic surfactants with different alkyl chain lengths on acid phosphatase, one of the lysosomal enzymes, and suggested that although inhibition and irritation is not related, surfactants with a certain level of irritating effect seem to have a marked inhibitory effect on acid phosphatase. Therefore, evaluation of the inhibitory ability by MAP-type of surfac- tants was also required for the clarification of the causative factors responsible for low irritation. Figure 22 exhibits initial rate-vs.-substrate concentration curves of acid phosphatase in the presence of the varying concentrations of C•2AS. Judging by both decreased Vmax and non-changed Km, C•2AS has a non-competive inhibitory effect on this enzyme. The inhibitory activity of MAP surfactants on acid phosphatase shown by percentage of inhibition plotted against the additive surfactant concentrations (Figure 23) indicates that C•SF and C•2MAP have a similar inhibitory ability to CuAS, whereas the effect of C•4, C•0 and CsMAP mono-sodium salts is at a significantly lower level (16). These findings indicate that, in general, these compounds possess almost the same inhibitory potential on acid phosphatase as common anionic surfactants except soap, indicating a similar order to protein denaturing effect described in SH releasing experiments. LYSOSOME LABILIZING EFFECT In addition to protein denaturation, the membrane damaging effect of surfactants may play an important role in evoking skin roughness or irritation (20,23). Since MAP is a phosphate derivative and the main component of cell or organelle membrane is phospholipid, it seems important to clarify if there is differential interaction with membrane between MAP and typical anionic surfactants like C•2AS. Therefore, the labilizing effect of these surfactants (10) was also investigated using subcellularly isolated lysosome-rich fraction from guinea pig skin (16). Thus, 1 ml of lysosome preparation which was obtained as supernatant fraction after centrifugation at 700 g • X 1• 5. g/ ml • rl• 5x 1• 4 • 150 '"1 O0 ........... ............. •0 Ci2AS C• 14• C10• C 12•P C 14• Na TEA TEA Figure 24. Labilizing effect of monoalkyl phosphates on guinea pig epidermal lysosome-rich fraction in comparison with C•2AS and C•2-14EO.
SKIN IRRITATION BY ANIONIC SURFACTANTS 63 =15 .. / ptt 8.5 /' //6 //•H 5.0 ß .-' C12 MAP monoTEA (5mM) o -"•" L-cell 0.146 mg/ml / i 6 0 120 180 Incubation Time fmin) Figure 25. Enzymic degradation of C•2MAP monoTEA salt in L-cell homogenate. for 10 min and at 12000 g for 10 min, was incubated at 37øC for 60 min with ! ml of each surfactant solution or ion-exchanged water as control. In order to evaluate the labilizing effect, the activity of lysosomal enzymes released after incubation was assayed according to the method of Shibko (21) for acid phosphatase in the supernatant obtained after centrifugation at 12000 g for 10 min. Figure 24 shows the labilizing effect of MAP on the lysosome fraction compared to C•2AS and C•2-14EO (16). With C•2AS , a release of acid phosphatase beyond control level was first found at 5 x 10-Sg/ml concentration. However, when a concentration of 5 x 10-4g/ml was employed, less acid phosphatase activity than that of control was observed, probably due to its inhibitory action. In the case of C•2-14EO , while the level of acid phosphatase activity released in the supernatant fraction was the same as control at the concentration of 5 x 10-Sg/ml, at 5 x 10-4g/ml the level rose beyond that of control. The labilizing data for various MAP's indicate that the induction of labilization requires a surfactant concentration of more than 5 x 10-4g/mi. These 20 Incubation Hours 5mM Ci2MAPmonoTEA Acetate Buffer pH5.0 Guinea Pig Epidermis Homogenate Figure 26. Enzymic degradation of C•2MAP monoTEA salt in epidermal homogenate of guinea pig skin.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)