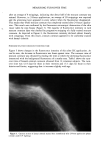
312 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS lOO 90 80 70 60 50 40 30 20 10 0 I i I I I I I I i I i I i I i I 2 4 6 8 10 12 14 16 18 MONTHS ON STABILITY TEST Figure 4. D-values for S. aureus in shampoo after storage at 3 ø, 20 ø, 38 ø, and 49øC for 1, 3, 6, 12, and 18 ø C mo. Explanation of symbols: &--&, shampoo stored at 49øC O--O, shampoo stored at 38 I--I, shampoo stored at 20øC and }--{, shampoo stored at 3øC. Ea' values obtained with S. aureus were calculated to be -2 and -4 Kcal/mole from D-values obtained after the shampoo was stored for 6 and 12 mo. respectively. Regression lines for the plot of Ea' vs months of storage were calculated for the test organisms (Figure 7). The slopes of these regressions showed a similar progression for all of the test organisms, exhibiting values of - 0.8, - 0.4, - 0.6, and - 0.3 Kcal/mole/ month and the corresponding correlation coefficients (r) of -1.00, -0.93, -0.75, and - 1.00 for E. coli, P. aeruginosa, Bacillus sp., and S. aureus, respectively. These data suggest that the loss of preservative system potency in the shampoo followed first-order or pseudo first-order reaction kinetics. DISCUSSION Accelerated aging, or storage at elevated temperatures, has been used to study the shelf life of cosmetic products and/or to determine the effect of physical and chemical param- eters of the formula on preservative stability (8,9). Several reports indicate that cosmetic preservative systems may interact with components of the formula or packaging mate- rials, with concomitant loss of preservative potency (9-14). In the present study, the preservative system of the shampoo was found to be unstable, as indicated by the change in D-values with time and temperature. Higher storage temperatures produced fairly rapid deterioration in shampoo preservation, as deter- mined with E. coli, P. aeruginosa, and Bacillus sp. This inactivation of the preservative system was not as obvious when using S. aureus as the challenge organism (Figure 4) because the net antibacterial effect of the shampoo (discussed below) inactivated S.
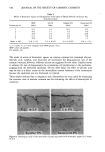
SHAMPOO PRESERVATIVE TESTING 313 Figure 5. HPLC chromatogram of shampoo spiked with methylparaben (MP), chloromethylisothiazo- linone (CMIT), and methylisothiazolinone (MIT). aureus rapidly. The differences in the rates of inactivation of the preservative system occurring at 3 ø to 49øC were not due to differences in the shampoo samples, because all samples used at each time/temperature period came from the same bottle. The shampoo stored at 49øC for over one month was the least resistant to contamination by E. coli, as evidenced by the larger D-values obtained in comparison with those observed when using the other test organisms. This may be due, in part, to limited activity of MP against gram-negative organisms (2) and, in part, to the ability of coli- forms to grow in the presence of anionic surfactants. For example, 0.0 ! % sodium lauryl sulfate is used to increase the selectivity for coliforms in enrichment media such as Lauryl Tryprose Broth (16). These findings show that different microorganisms do not respond monotonically to the preservative system in the shampoo. This is why it is essential that preservative efficacy testing be performed using test organisms with all of the physiological characteristics expected to be a potential problem in the formula. As a minimum, gram-negative organisms with diverse metabolic capabilities, such as P. aeruginosa, a representative of the coliform group, such as E. coli, a gram-positive coccus, such as S. aureus, and a gram-positive, spore-forming rod (i.e., Bacillus sp.) should be included in preservative testing. These bacteria, in addition to selected yeasts and/or molds not discussed in the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)