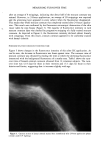
314 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Figure 6. HPLC chromatogram of shampoo after storage at 3øC for 18 months. current work, provide sufficiently diverse morphological and physiological character- istics to give reasonable assurance that the test results offer a good indication of the resistance of the product to contaminants that could come into contact with the product during production or while in the hands of the consumer. Other considerations in the selection of test organisms have been discussed (2). It is known that some cosmetic raw materials affect preservative efficacy. In addition, it should be recognized that the preservative system of a cosmetic product may involve Table I Change in Apparent Activation Energies (Ea') of Shampoo During Stability Testing Ea' (Kcal/mole)* Test Organism 0 mo 1 mo 3 mo 6 mo 12 mo E. coli -** -7 -9 - 1! - 16 P. aeruginosa - - 6 - 6 - 9 - 10 Bacillus sp. - - 4 - 3 - 10 - 9 S. aureus .... 2 - 4 * Calculated by substitution of 2. 303/D-value for k in Arrhenius equation (see equation {3}). ** Calculation not performed due to lack of sufficient data.
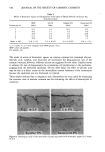
SHAMPOO PRESERVATIVE TESTING 315 o -1 -2 -3 -4. -5 -6 -7 -8 -9 -lO 11 -12 -13 -14. -15 -16 -17 -18, -19 -20 o 1 2 3 4 5 6 7 • 9 lo 11 12 13 14 MONTHS ON STABIUTY TEST Figure 7. Change in apparent activation energies (Ea') for the shampoo preservative system during the stability study, determined using test organisms. Explanation of symbols: I--I, E. coli O--O, P. aeruginosa •--•, Bacillus sp. and l•__,, S. aureus. more than chemicals with known antimicrobial activity. Factors such as pH, water activity, nutrient availability, surfactant concentration, sequestering agents, and other interferences will determine the extent to which preservative action is manifested in any given formulation. Thus, the preservative system of a product involves both specific preservative chemicals and the physicochemical constitution of the entire formulation. The inhibition of bacterial growth in a shampoo may conceivably be due to more than one mechanism (i.e., surfactant destabilization of cell membranes, preservative action on cellular metabolism, sequestration of divalent metal ions by tetrasodium EDTA, unavailability of nutrients, etc.). One would not expect organisms with different meta- bolic capabilities to be inactivated at the same rate in any cosmetic product. In the current study, it was found that the four test organisms responded differently to the net antibacterial effect of the shampoo. Although the goal of this investigation was not to determine the cause of change in preservative efficacy, the HPLC results revealed the presence of all three preservatives initially, but only MP was unchanged after one week of storage at room temperature (Figures 5 and 6). This suggests that CMIT and/or MIT reacted with some component in the formula--possibly hydrolyzed animal collagen, because these isothiazolinones are known to react with amines (15). It is known that the rate constants for chemical reactions are influenced by temperature, as represented by the Arrhenius equation (17) k = Ae -r'•RT where k is the reaction rate constant, A is the pre-exponential factor, Ea is the activa- tion energy, R is the gas constant, and T is the absolute temperature. Differentiating
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)