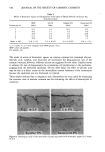
316 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS equation (1} with respect to temperature and integrating between limits yields equation {2} (17): log k2 - Ea (T2 - T,) {2} k• 2.303R (T•T 2) Stumbo (7) reported that the D-value = 2.303/k consequently, k = 2.303/D-value. Substituting 2.303/D-values for k enables one to use an expression that relates D- values, Ea and T, as in equation {3}: D-value• _ Ea (T 2 - T•) {3} log D-value2 2.303R (T•T 2) The rates of reactions in biological systems are affected similarly by temperature, so that the rate of inactivation of a given organism in the presence of preservative chemicals (or other physicochemical conditions that are bacteriocidal) increases with temperature. Since D-values become smaller as the rates of microbial inactivation increase, it would be expected that the D-value for a given organism in a test sample would decrease with an increase in preservative efficacy test temperature--as long as the preservative system was not altered by the assay conditions. The situation is different in the current study since the shampoo preservative potency was evaluated at constant conditions (i.e., by performing all preservative efficacy tests at room temperature) after test samples had been stored for specified times at different temperatures. The preservative system was found to be unstable when tested by the linear regression method. This decrease in preservative potency with time and tempera- ture of storage resulted in decreases in the slopes of the survivor curves (4) and corre- sponding increases in D-values for each test organism. Although the slope of the Arrhenius activation energy plot is negative when k increases with temperature, the decrease in rates of bacterial inactivation with temperature ob- served in this study gives a positive slope in the Arrhenius activation energy plot, as is illustrated for the data obtained with the test samples stored for 12 mo. and challenged with E. coli (Figure 8). The Ea calculated from these results are negative consequently, they are designated Ea'. The Ea' values calculated for shampoo preservative potency during the first 12 mo. of the stability study appear in Table I. The progressive decrease in Ea' for all test or- ganisms reflects the decrease in preservative system potency. Here, the rate of change of preservative system potency was greatest for E. coli (i.e., the organism most resistant to the shampoo preservative system) and smallest for S. aureus (the test organism least resistant to the preservative system). Although the negative Ea' values appear to be contradictory to conventional systems in which Ea is determined, one should note that the parameter being measured--preser- vative system potency--decreased with increasing temperature, as determined by the kinetics of inactivation of the test organisms. In general, the rate of a chemical reaction, as expressed by k, is a function of the concentration of the reactants. If the concentration of a reactant (i.e., preservative) is changed as a result of storage at elevated temperatures for different times, determining D-values and using 2.303/D-value enables one to determine k at different tempera- tures. These k values may be compared with k values obtained in systems of known
SHAMPOO PRESERVATIVE TESTING 317 I i r 3.0 3.2 3.4 3.6 1000/T Figure 8. Arrhenius activation energy plot of shampoo preservative system potency determined using E. coli as the challenge organism in preservative efficacy tests on samples stored at 3 ø, 38 ø, and 49øC for 12 months. preservative molar concentration, so that one may determine the molar concentration (or the apparent molar concentration) of active preservative in the formula after any given time and temperature of storage. Calculations of this type may be useful in systems containing only one preservative chemical. Obviously, calculations will be quite complex in products containing multiple surfactants and preservative chemicals because the loss of preservative potency may follow higher-order reaction kinetics. It is believed that determining D-values and Ea' values may be useful in studying the ki- netics of bacterial death, in determining the apparent molar concentration of a preser- vative, and in monitoring the performance of cosmetic preservative systems during stability studies. In discussing accelerated stability testing, Pope used Ea values of 10-20 Kcal/mole for predicting good probability of formulation stability, which he defined as one that de- graded no more than 10.5% in 3 mo. at 45øC (18). He noted that formulations that degrade through solvolysis have Ea values of 10-30 Kcal/mole and that systems with Ea values of this magnitude show marked increases in reaction rates at elevated tempera- tures. The findings in this study revealed that the unstable shampoo preservative system had Ea' values of - 2 to - 16 Kcal/mole, depending on the time period of the determi- nation and the test organism used. It is believed that the difference between the Ea values proposed by Pope and the Ea' values observed in this study (including both absolute magnitude and algebraic sign) may be due to 1) the difference in k for the solvolysis reactions cited by Pope and complexation reactions, such as those involved in the interaction of isothiazolinones and amines (15) and 2) the way in which Ea and Ea' were defined and derived. The shampoo preservative system was satisfactory when examined at the outset of these studies (i.e., at 0 mo.), but it deteriorated during the aging study. Hence, the Ea'
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)