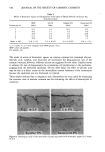
338 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 50- 40 • 30 c• 20 10 I O0 - 24 48 72 Time (h) Figure 5. Cumulative amount of EPC-Na permeated across the skin. The circles and error bars represent the average and standard error of six data points. steady-state rate of permeation of EPC-Na and the steady-state rate of appearance of vitamin C, the yield of bioconversion was calculated to be 96% in the hairless mouse skin. It is obvious, however, that the yield of the provitamin bioconversion is in- fluenced by the activity and distribution of enzymes in the skin therefore, the findings obtained from the hairless mouse may not be directly applicable to man. Suffice it to say, however, that the provitamin, EPC-Na, will be bioconvertible to vitamins C and E simultaneously in the human skin because of the enzyme (esterase) distribution in human skin (17). ACKNOWLEDGEMENTS We express our sincere thanks to Senju Pharmaceutical Co. Ltd., Osaka, Japan, for providing EPC-Na. We are also grateful to Drs. Y. W. Chien and O. Siddiqui for their valuable comments. REFERENCES (1) L. J. Machlin, in Handbook of Vitamins (Marcel Dekker, New York, 1984). (2) C. J. Bates, in Vitamin C (Astorbit Acid), J. N. Counsell and D. H. Hornig, Eds. (Applied Science Publishers, Englewood, NJ, 1982), pp. 1-22. (3) D. Djerassi, Vitamin E: Biochemical function and its role in cosmetics, D&CI, 46-77 (March 1986). (4) K. Furuse, Vitamin E--Biochemistry and biochemical function, Pharm. Tech. Japan, 2, 729-735 (1986). (5) R. E. Keith, B. M. Chrisley, and J. A. Driskell, Dietary vitamin C supplementation and plasma vitamin E levels in humans, Am. J. Clin. Nutr., 33, 2394-2395 (1980). (6) E. Ginter, A. Kosinova, A. Hudecova, and A. Madaric, Synergism between vitamins C and E: Effect on microsomal hydroxylation in guinea pig liver, Internat. J. Vit. Nutr. Res., 52, 55-59 (1982). (7) L. J. Machlin, in Vitamin E (Marcel Dekker, New York, 1980).
PROVITAMIN TO C AND E IN SKIN 339 (8) (9) (10) (11) (12) (13) (14) (15) (16) (17) (18) (19) M. A. Kassem, A. A. Kassem, and H. O. Aremar, Studies on the stability of injectable L-ascorbic acid solutions, Pharm. Acta Helv., 44, 611-623 (1969). Y. W. Chien, Logics of transdermal controlled drug administration, Drug Dev. & Ind. Pharm., 9, 497-520 (1983). J. Shaw, Development of transdermal therapeutic systems, Drug Dev. & Ind, Pharm., 9, 579-604 (1983). W. I. Higuchi, N. A. Gordon, J. L. Fox, and N. F. H. Ho, Tarnsdermal delivery of prodrugs, Drug Dev. & Ind. Pharm., 9, 691-706 (1983). Y. Ishida, Action of vitamin C and its derivatives, FragranceJ. (Japan), 63, 28-34 (1983). T. Nisiyama, M. Naganuma, Y. Fujinuma, and K. Nakajima, Studies on the percutaneous absorp- tion and the skin metabolism of ascorbic acid 2-sulfate in guinea pigs, Vitamins (Japan), 56, 537-542 (1982). T. Shiratori, Uptake, storage and excretion of chylomicra-bound 3H-a-tocopherol by the skin of the rat, Life Sciences, 14, 929-935 (1974). K. Tojo, J. A. Masi, and Y. W. Chien, Hydrodynamic characteristics of an in vitro drug permeation cell, I&EC Fundamentals, 24, 368-373 (1985). K. H. Valia, K. Tojo, and Y. W. Chien, Long term permeation kinetics of estradiol: (III) Kinetic analyses of the simultaneous skin permeation and bioconversion of estradiol esters, Drug Dev. Ind. Pharm., 11, 1133-1173 (1985). Y. Miura, Histochemical studies on enzyme activities in the skin, J. Dermatol. Japan (in Japanese), 74, 556-570 (1964). B. D. Bunday, in Basic Optimisation Method (Edward Arnold Pub., 1984), Part 4, pp. 47-76. E. Ginter and P. Bobek, in Vitamin C, J. N. Counsell and D. H. Hornig Eds. (Applied Science Publishers, Englewood, NJ, 1981), pp. 299-347.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)