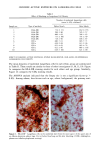
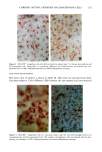
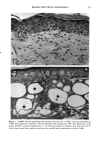
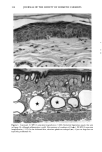
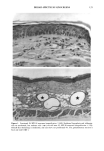
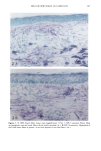
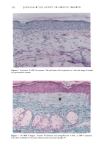
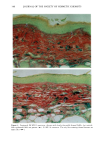
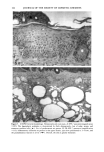
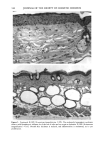
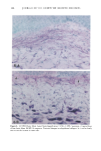
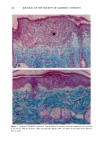
126 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS mean age of these two groups. Gilhar et al. reported that the number of Langerhans cells in an aged group was approximately 50% of that in a younger group (13). Gilchrest et al. also reported a decrease of Langerhans cells in an aged group (4). A relationship between age-associated decrease of Langerhans cell density and deficiency in bone mar- row progenitors was suggested from the results of the transplantation of bone marrow cells in mice (13). Such a decrease would be reasonable in the light of generally decreasing immunological function with advancing age (21). However, our study did not show such a dramatic decrease but gave an equivocal result. The discrepancy between our study and Gilhar's study may be attributable to the difference in the age of the older age groups. The mean age of the older subject group in Gilhar's study was 82.1 years, which is considerably higher than that of our subject groups (Caucasian 66.0, African-American 69.6). On the other hand, the mean age of the older group in the report of Gilchrest et al. (4), 69, was almost the same as that of the African-American aged group in our study. In our study, the Langerhans cell density of the African-American elderly group is slightly lower than those of other groups. Thus, we consider that Langerhans cell density may start to decrease at around 70 years of age, and that the discrepancy between the two ethnic groups is not due to ethnic background but to the slight difference in the age of the two older groups. In other words, Langerhans cell density is maintained at the normal level until the eighth decade of life in healthy individuals, and the age of 66.0 is not old enough to show the decrease of Langerhans cell density. The primary antibody used for staining was also a statistically significant factor. This result most likely suggests that Langerhans cells in epidermis consist of two different subsets in phenotype, possibly a mature type and an immature type, but that these two subsets are considered to be of the same lineage. Harrist et al. reported that the number of CDla-positive Langerhans cells was approximately 2- to 3-fold the number of HLA- DR-positive Langerhans cells (22). Fithian et al. demonstrated that approximately 90% of CDla-positive cells were HLA-DR positive by immunofluoresence double staining (23). We cannot reach a definitive conclusion since double staining was not performed in our study. However, the number of HLA-DR positive cells was approximately 95% of that of CDla-positive cells. This result is comparable to that of Fithian et al. (23). Although two different subsets (CDla +/HLA-DR + and CDla +/HLA-DR-) may exist, our conclusion would not be affected since they showed the same tendencies. Our preliminary experiment showed that a small amount of acute UVB radiation, which simulated the sunlight exposure on a daily basis, differently damaged the Langerhans cell density of two ethnic groups possibly because of the protective effect of melanin pigmentation (data not shown). Therefore, before starting the study, we expected that the effect of chronic actinic exposure on Langerhans cell density might differ in these two groups. However, the effect of chronic actinic exposure was not apparent in this study. It is reasonable that there would not be any differences associated with ethnic back- ground if there is no chronic low-level UV effect. Our results indicate that Langerhans cell density, even at sun-exposed sites, is preserved as long as the systemic immune function is maintained in a normal condition, since Langerhans cells derive from bone marrow (24). We conclude that there is no identified chronic sunlight effect and no identified dif- ference between the two ethnic groups with respect to Langerhans cell density, and also
CHRONIC ACTINIC EXPOSURE ON LANGERHANS CELLS 127 that Langerhans cell density starts to decrease late in life, after 70 years of age. Our observations, however, do not extend to high-level chronic UV exposures such as those occurring in outdoor workers. In such populations at high risk of developing UV- induced skin cancers, the possible protective effect of pigmentation against the actinic damage of Langerhans cells remains to be determined. More importantly, as the sun- screens in cosmetics are reported to protect Langerhans cells from acute photodamage (25), the use of sunscreens is also beneficial to prevent the cumulative sunlight damage that may cause secondary damage to Langerhans cells in the later stage of life. ACKNOWLEDGMENTS The authors would like to thank Dr. K. L. Gabriel and Dr. R. Mark at Biosearch, Incorporated for their help in completing the study. Financial support from Biosearch, Incorporated is gratefully acknowledged. REFERENCES (9) (10) (11) (12) (13) (14) (15) (16) (1) I. Silberberg-Sinakin, G. J. Thorbecke, S. A. Rosenthal, and V. Berezowsky, Antigen-bearing Lang- erhans cells in skin, dermal lymphatics and in lymph nodes, Cell. Immunol., 25, 137-151 (1976). (2) W. Aberer, G. Schuler, G. Stingl, H. Honingsmann, and K. Wolff, Ultraviolet light depletes surface markers of Langerhans cells, J. Invest. Dermatol., 76, 202-210 (1981). (3) O. Baadsgaard, K. D. Cooper, S. Lisby, H. C. Wulf, and G. L. Wantzin, Dose response and time course for induction of T6-DR + human epidermal antigen-presenting cells by in vivo ultraviolet A, B, and C irradiation, J. Am. Acad. Dermatol., 17, 792-800 (1987). (4) B. A. Gilchrest, G. F. Murphy, and N. A. Soter, Effect of chronologic aging and ultraviolet irra- diation on Langerhans cells in human epidermis, J. Invest. Dermatol., 79, 85-89 (1982). (5) J. Alcalay, L. H. Goldberg, J. E. Wolf, and M. L. Kripke, Ultraviolet radiation-induced damage to human Langerhans cells in vivo is not reversed by ultraviolet A or visible light,J. Invest. Dermatol., 95, 144-146 (1990). (6) D. V. Belsito, R. L. Baer, I. Gigli, and G. J. Thorbecke, Effect of combined topical glucocorticoids and ultraviolet B irradiation on epidermal Langerhans cells,J. Invest. Der matol., 83, 347-351 (1984). (7) M. Obata and H. Tagami, Alteration in murine epidermal Langerhans cell population by various UV irradiations: Quantitative and morphologic studies on the effects of various wavelengths of mono- chromatic radiation on Ia-bearing cells, J. Invest. Derm•tol., 84, 139-145 (1985). (8) J. Czernielewski, P. Vaigot, D. Asselineau, and M. Prunieras, In vitro effect of UV radiation on immune function and membrane markers of human Langerhans cells, J. Invest. Der matol., 83, 62-65 (1984). J. Austad and L. R. Braathen, Effect of UVB on alloactivating and antigen-presenting capacity of human epidermal Langerhans cells, Scand. J. Immunol., 21, 417•i23 (1985). B. H. Thiers, J. C. Maize, S. S. Spicer, and A. B. Cantor, The effect of aging and chronic sun exposure on human Langerhans cell population, J. Invest. Dermatol., 82, 223-226 (1984). B. A. Gilchrest, G. Szabo, E. Flynn, and R. M. Goldwyn, Chronologic and actinically induced aging in human facial skin, J. Invest. DermatoL, 80, 81s-85s (1983). V. A. Delo, L. Dawes, and R. Jackson, Density of Langerhans cells in normal vs. chronic actinically damaged skin of humans, J. Invest. Dermatol., 76, 330-331 (1981). A. Gilhar, T. Pillar, M. David, and S. Eidelman, Melanocytes and Langerhans cells in aged versus young skin before and after transplantation onto nude mice, J. Invest. Dermatol., 96, 210-214 (1991). D. N. Sauder, Effect of age on epidermal immune function, Dermatol. C/in., 4, 447-454 (1986). E. Sprecher, Y. Becker, G. Kraal, E. Hall, D. Harrison, and L. D. Shultz, Effect of aging on C57BL/6J mice, J. Invest. Dermatol., 94, 247-253 (1990). B. Berman, V. L. Chen, D. S. France, W. I. Dotz, and G. Petroni, Anatomical mapping of epi- dermal Langerhans cell densities in adults, Br. J. Dermatol., 109, 553-558 (1983).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)