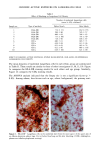
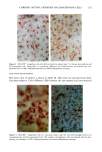
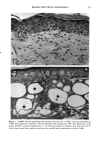
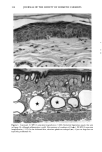
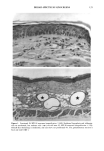
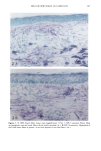
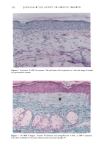
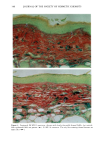
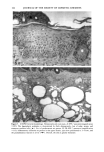
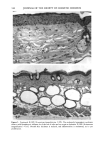
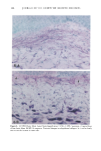
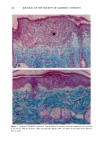
128 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (17) R. D. Sontheimer, P. Stastny, and G. Nunez, HLA-D region antigen expression by human epidermal Langerhans cells, J. Invest. Dermatol., 87, 707-710 (1986). (18) J. L. Cordell, B. Falinia, W. N. Erber, A. K. Ghosh, A. Abdulaziz, S. MacDonald, K. A. F. Pulford, H. Stein, and D. Y. Mason, Immunoenzymatic labeling of monoclonal antibodies using immune complexes of alkaline phosphatase and monoclonal anti-alkaline phosphatase (APAAP com- plexes),J. Histochem. Cytochem., 32, 219-229 (1984). (19) H. Chen, J. Yuan, Y. Wang, and W. K. Silvers, Distribution of ATPase-positive Langerhans cells in normal adult human skin, Br. J. Dermatol., 113, 707-711 (1985). (20) M. Hatao, R. Mark, T. Stoudemayer, and K. L. Gabriel, Recovery process of Langerhans cells in human skin following UVB irradiation,.]. Toxicol. Cut. Ocul. Toxicol., 12, 293-301 (1993). (21) T. Makinodan, Immunobiology of aging, J. Am. Geriat. Soc., 24, 249-252 (1976). (22) T. J. Harrist, J. E. Muhlbauer, G. F. Murphy, M. C. Mihm, Jr., and A. K. Bhan, T6 is superior to Ia (HLA-DR) as a marker for Langerhans cells and indeterminate cells in normal epidermis: A monoclonal antibody study, J. Invest. Dermatol., 80, 100-103 (1983). (23) E. Fithian, P. Kung, G. Goldstein, M. Rubenfeld, C. Fenoglio, and R. Edelson, Reactivity of Langerhans cells with hybridoma antibody, Proc. Natl. Acad. Sci. USA, 78, 2541-2548 (1981). (24) S. I. Katz, K. Tamaki, and D. H. Sachs, Epidermal Langerhans cells are derived from cells origi- nating in bone marrow, Nature, 282, 324-326 (1979). (25) C. A. Elmets, A. Vargas, and C. Oresajo, Photoprotective effects of sunscreens in cosmetics on sunburn and Langerhans cell photodamage, Photodermatol. Photoimmunol. Photomed., 9, 113-120 (1992).
j. Soc. Cosmet. Chem., 47, 129-155 (May/June 1996) Broad-spectrum sunscreens with UVAI and UVA II absorbers provide increased protection against solar-simulating radiation-induced dermal damage in hairless mice LORRAINE H. KLIGMAN, Department of Dermatology, University of Pennsylvania School of Medicine, Philadelphia, PA 19104-6142, PATRICIA P. AGIN, Research and Development, Schering-Plough Health Care Products, 3030Jackson Avenue, Memphis, TN 38151, and ROBERT M. SAYRE, Rapid Precision Testing Laboratory, Cordova, TN 38018-1342. Accepted jSr publication June 19, 1996. Synopsis Previous experiments designed to examine sunscreen protection against chronic UV radiation-induced skin damage in hairless mice have used radiation sources emitting mainly UVB or UVA radiation. Because humans are exposed to full-spectrum solar radiation, we were interested in examining the efficacy of three sunscreens, with increasing spectral absorption into the UVA range, against chronic solar-simulating radiation (SSR). Three groups of hairless mice received a cumulative SSR dose of 10 and 16 times a previously determined minimal photoaging dose (MPD) over periods of 18 and 30 weeks. Each twice- weekly exposure was designed to equal the SPF value of the first sunscreen, an SPF-7 sunscreen containing the UVB absorber octyl methoxycinnamate. The second sunscreen, in addition to the UVB absorber, contained a UVA II absorber (oxybenzone) and had an SPF of 16. The third, with an SPF of 18, contained the UVB and UVA II absorbers plus a UVA I absorber (avobenzone). These conditions allowed assessment of the effects of UVB and UVA radiation that are normally transmitted through all sunscreens. Although none of the sunscreen-treated mice developed erythema, considerable dermal matrix damage occurred in the SPF-7 group, with greater damage at 16 MPD than at 10 MPD. The SPF-16 sunscreen allowed less but clearly recognizable damage at both dose points. The SPF-18 sunscreen with the broadest spectral absorp- tion provided the greatest protection. These results support the need for high-SPF broad-spectrum sun- screen protection that includes the entire UVA spectrum to reduce photodamage that results from chronic exposure to sunlight. INTRODUCTION The hairless mouse has proved a useful model for demonstrating the protective effect of sunscreens against the diverse consequences of chronic ultraviolet radiation. For exam- ple, sunscreens with high sun protection factors (SPF) have been shown to prevent UVB-induced photocarcinogenesis (1,2) and the dermal connective tissue damage char- 129
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)