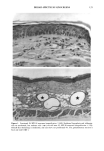
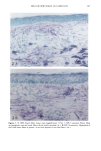
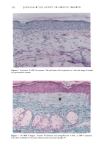
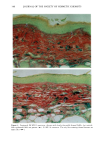
PHOTOTOXICITY TESTING 17 5 (9) L. C. Harber, A.M. Lashinsky, and R. L. Baer, Photosensitivity due to chlorothiazide and hydro- chlorothiazide, N. Engl. J. Med., 261, 1378-1381 (1959). (10) H. Schatz, M. Mark, and H. P. T. Ammon, Antidiabetika (Wissenschaftliche Verlagsgesellschaft, Stuttgart, 1986). (11) R. Diising, Diuretika (Wissenschaftliche Verlagsgesellschaft, Stuttgart, 1986). (12) J. Moan, B. H0vik, and S. Sommer, A device to determine fluence-response curves for photoinacti- vation of cells in vitro, Photochem. Photobiophys., 8, 11-17 (1984). (13) K. Nordbye and R. Oftebro, Establishment of four new cell strains from human uterine cervix, J. Exp. Cell Res., 58, 458 (1969). (14) T. T. Puck, S. J. Cieciura, and H. Fisher, Clonal growth in vitro of human cells with fibroblastic morphology, J. Exp. Med., 106, 145-165 (1957). (15) I. Matsuo, H. Fujita, K. Hayakawa, and M. Ohkido, Lipid peroxidative potency of photosensitized thiazide diuretics, J. Invest. Dermatol., 87, 637-641 (1986). (16) H. Fujita and I. Matsuo, An in vitro test for photoinduced toxicity of benzothiadiazine diuretics using bacteriophage lambda, Photochem. Photobiol., 41, 355-359 ( 1985). (17) I.E. Kochevar, Mechanisms of drug photosensitization, Photochem. Photobiol., 45, 891-895 (1987). (18) M. L. Rooney, Ascorbic acid as a photooxidation inhibitor, Photochem. Photobiol., 38, 619-629 (1983). (19) J. Moan, On the diffusion length of singlet oxygen in cells and tissues, Photochem. Photobiol., 6, 343-344 (1990). (20) B. E. Johnson, E. M. Walker, and A.M. Hetherington, "In Vitro Models for Cutaneous Phototox- icity," in Skin Models, Marks, Piewig, Eds. (Springer Verlag, Berlin, Heidelberg, New York, 1986), pp. 264-281. (21) A. E. Ison and C. M. Davies, Phototoxicity ofquinoline methanols and other drugs in mice and yeast, J. Invest. Dermatol., 52, 193-198 (1969). (22) F. Daniels, A simple microbiological method for demonstrating phototoxic compounds, J. Invest. Dermatol., 44, 259-263 (1965). (23) T. Horio, Evaluation of drug phototoxicity by photosensitization of trichophyton mentagrophytes, Br. J. Dermatol., 105, 365-370 (1981). (24) W. L. Morison, D.J. McAuliffe, J. A. Parrish, and K.J. Bloch, In vitro assay for phototoxic chemicals, J. Invest. Dermatol., 78, 460-463 (1982). (25) R. G. Freeman, W. Murtishaw, and J. M. Knox, Tissue culture techniques in the study of cell photobiology and phototoxicity, J. Invest. Dermatol., 54, 164-169 (1970).
j. Soc. Cosmet. Chem., 47, 177-184 (May/June 1996) Rapid quantitative determination of 1,4-dioxane in cosmetics by gas chromatography/mass spectrometry DEMING SONG, SHIDE ZHANG, WENWEN ZHANG, and KENNETH KOHLHOF, Clinical Research Associates, 50 Madison Avenue, New York, NY, 10010. Accepted for publication June 19, 1996. Synopsis Dioxane is a potential carcinogen. Determination of the level of dioxane present in cosmetics is of impor- tance. A highly sensitive and accurate gas chromatography/mass spectrometry method for the rapid quan- titative analysis of dioxane in cosmetics is described. Dioxane was extracted from a sample by hexane and methylene chloride (80:20, v/v) and then transferred to a C 8 solid-phase extraction (SPE) cartridge and eluted by acetonitrile. An aliquot of this solution was analyzed by gas chromatography/mass spectrometry with electron impact ionization (El) and a selected ion monitoring (SIM) (dioxane m/z = 88) mode. The quantitative limit was set to be 0.1 ppm (0.1 •xg/g for cosmetics) with an injection volume of 2 ptl. The isotopically labeled dioxane-d 8 (m/z = 96) was used as an internal standard. INTRODUCTION 1,4-Dioxane is an anticipated carcinogen (1,2) and a possible by-product in the pro- duction of polyethoxylated surfactants that are widely used in cosmetic emulsions such as lotions and shampoos (3,4). 1,4-Dioxane could be formed during the polymerization of ethylene oxide to produce the polyoxyethylene portion of the emulsifiers (5). Dioxane is water-soluble and has a boiling point of 10 IøC. Accordingly, once dioxane forms in water-soluble cosmetics, it is not easily eliminated. Hence the potential problems associated with 1,4-dioxane have been brought to the attention of the cosmetic industry, and 1,4-dioxane has been the object of tests in various kinds of cosmetic products (6-9). Several methods of GC (6,7), HPLC (8), and GC/MS (9) have been presented for the testing of 1,4-dioxane in cosmetics. These methods have their limitations. GC or GC/MS methods suffer from unsatisfactorily low accuracy and reproducibility (6). The HPLC method suffers from low sensitivity: 6.5 ppm (p•g/g) (8). In comparison, our method provides a higher sensitivity (0.1 ppm) and shorter analysis time (6 min) than any of the present methods. Moreover, the use of the isotopically labeled compound dioxane-d 8 as an internal standard in our method has greatly improved the accuracy and reproducibility in both intra-assay and inter-assay. The structures of dioxane and diox- ane-d 8 are shown in Figure 1. 177
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)