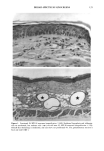
178 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS / \ D 1,4- DIOXANE 1,4- DIOXANE-D 8 M.W.=88.1 M.W.=96.1 Figure 1. The structures of 1,4-dioxane and internal standard dioxane-d 8. EXPERIMENTAL CHEMICALS AND MATERIALS 1,4-Dioxane, 99.9%, HPLC grade, and 1,4-dioxane-d 8, 98.5 atom % of D, were purchased from Sigma (St. Louis, MO). Dichloromethane and acetonitrile HPLC grades were purchased from EM Industries, Inc. (Gibbstowne, NJ). Hexane, HPLC grade, and solid phase extraction cartridge C 8 were the product of J. T. Baker Chemical Co. (Phillipsburg, NJ). SAMPLE PREPARATION A cosmetic product (about 0.4 g) was accurately weighed into a 15-ml glass test tube. Fifty microliters of internal standard working solution (200 ppm) and enough water were added to make the total volume 1 mi. Then 6 ml hexane:dichloromethane (80:20, v/v) solution was added. The test tube was capped and vortexed for 15 minutes. The sample was then centrifuged for ten minutes at ambient temperature at 700 g. The extraction solution layer was pipetted to a SPE C 8 cartridge that was preconditioned prior to use by 1 ml of acetonitrile and 1 ml of extraction solution. The SPE C 8 tube was then centrifuged again at 700 g for five minutes and eluted by 0.5 ml acetonitrile two times. An aliquot (50 •1) of the elution solution was transferred to a sample vial for analysis by GC-MS. METHOD OF ANALYSIS Analysis was performed on an HP 5890 II gas chromatograph with an HP-5 column (30 m x 0.25 mm ID, 0.25-•m film thickness) and an HP 5989 MS engine equipped with a Model 7673 autosampler (model HP DOS G1034C MS ChemStation, Hewlett Pack- ard, Palo Alto, CA). Helium was the GC carrier gas, with a column head pressure of 30 KPa at about 25øC, and the flow rate was maintained at 1 ml/min. The injector temperature was 130øC. The
DETERMINATION OF DIOXANE LEVELS 179 oven temperature programming started at an initial temperature of 40øC for four min- utes, then raised at a rate of 10øC/min to 100øC. The injection volume was 2 •1. The running time was approximately six minutes. The mass was set at the E1 and SIM acquisition mode: monitoring ions at m/z 88 and 96. The temperatures of source and quadrupoles were 150øC and 100øC, respectively. The ionizing electrons and electrical multiplier (EM) were set at 70 eV and 2000V, respectively. CALIBRATION STANDARD CURVE AND QUANTITATIVE ANALYSIS A standard sample for calibration curve was prepared, in duplicate, in a test tube, by adding 50 •1 of the respective working solution (containing the analyte: 0.1, 0.5, 5.0, 25.0, 50.0, and 100.0 •g), 50 •1 of the internal standard working solution (containing the internal standard 10.0 •g), and deionized water to a total volume of 1 mi. The final concentrations of six dioxane standards were 0.1, 0.5, 5.0, 25.0, 50.0, and 100.0 ppm (•g/ml), respectively. The concentration of internal standard dioxane-d 8 was 10.0 ppm. Quality control (QC) samples were run in duplicate at three different concentration levels: QC L = 1.0, QC M = 10.0, QC H = 75.0 ppm. The quantitative analysis was based on the ratio of the peak height of the analyte to that of the internal standard. RESULTS AND DISCUSSION SAMPLE EXTRACTION AND RECOVERY Acetonitrile was chosen as solvent for dioxane because of the following two points: First, the molecular weight ofacetonitrile (m.w. 44) is smaller than that of dioxane (m.w. 88). This is good for GC analysis since the big solvent peak may go out faster than that of the analyte and can be eliminated by a solvent delay program. Second, the dioxane has the best solubility in acetonitrile because of their close polarities (10). However, ace- tonitrile can not extract dioxane from most cosmetics (water-soluble and those contain- ing water) because it is miscible with water. On the other hand, hexane and methylene chloride have stronger extracting ability, but their molecular weights (m.w. 84 and m.w. 85, respectively) are too close to that of dioxane. The use of hexane or methylene chloride as a solvent for GC/MS will result in interference in the analysis. This problem is solved by the solid-phase extraction (SPE). By SPE, the dioxane extracted from cosmetics by hexane and methylene chloride is transferred to a solid-phase cartridge, and then dioxane is washed from the cartridge and eluted by acetonitrile. Therefore, ace- tonitrile is the final solvent for dioxane for GC analysis. Our experiments show that a number of SPE columns such as C2, C 8, C18 , NH2, and SiOH bonded to silica can do this job. In our work the C 8 cartridge by J. T. Baker Inc. was used as the SPE column. The recovery was found to be 86.7% (n = 6), calculated by the ratio of the peak height of the same standard sample with and without solid phase extraction to that of the internal standard without extraction in both cases. ISOTOPICALLY LABELED INTERNAL STANDARD AND REPRODUCIBILITY The serious problem for the analysis of dioxane by GC or GC/MS techniques is low accuracy and reproducibility as well as high variability in the recovery from different
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)