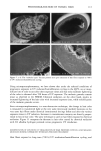
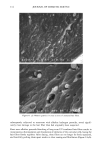
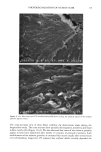
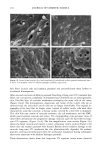
92 JOURNAL OF COSMETIC SCIENCE ending). The original concept, while creative, does not give the total information needed for defining the molecular structure. For example, the current practice is to call me- thoxy-capped products DMC, even though they lack the hydroxyl group that originally justified the "o1" ending. Since the capping process is not totally efficient, there are residual hydroxyl groups found even in the DMC compounds that claim to be capped. TERMINAL GROUP FUNCTIONALITY The structure of the DMC having a terminal group is shown below. For hydroxy- terminated DMC the structure is: CH 3 CH 3 CH 3 CH 3 CH3-Si--(-O--Si--)•-- ( O--Si)b--O--Si--CH 3 CH 3 CH 3 (CH2) 3 CH 3 I O-(CH2CH20)x-(CH2CH(CH3)O)yH For methoxy-terminated DMC the structure is: CH 3 CH 3 CH 3 CH 3 CH 3-Si--(-O--Si--)•--( O--Si)b--O--Si--CH 3 CH 3 CH 3 (CH2) 3 CH 3 I O-(CH2CH20)x-(CH2CH(CH3)O)yCH 3 The molecular structure is complicated further by the fact that the a, b, x, and y values vary quite considerably within the class and are generally not revealed by manufacturers. The raw materials used to synthesize these polymers are themselves polydisperse poly- mers having an oligomeric distribution. The resulting polymer is a complex oligomeric distribution of the initial oligomeric distribution. Despite the complications, which are not too unlike those found in ethoxylated fatty surfactants, silicone polymers can be analyzed and structure/function properties determined. The process used for their syn- thesis is reproducible and gives products with little variation, albeit complex mixtures (1•). The general reaction scheme for the synthesis of DMCs is as follows: CH 3 CH 3 CH 3 CH 3 CH3---Si---(-O---Si---)•---(-O--Si---)b--O--Si--CH3 CH 3 H CH 3 CH 3 + "a" moles of CH2=CH-CH2-O-(CH2CH20)x-H
DIMETHICONE COPOLYOL 93 CH 3 CH 3 CH 3 CH 3 CH 3---Si---(-O---Si---)a---(-O--Si---)b--O--Si--CH 3 CH 3 (CH2) 3 CH 3 CH 3 I O-(CH2CH20)x-H It must be clearly understood that the polymer structures shown above are equilibrium mixtures having a nominal or average structure. This is not unlike ethoxylated alcohols that are also oligomeric mixtures of different ethoxylated isomers (4). NOMENCLATURE In order to better understand the polymer chemistry, a shorthand has been developed that is more enlightening to the chemist than the name DMC. Developed in the 1940s by Dow Corning scientists, the nomenclature is based upon the type of groups present in the molecule. CH 3 I "M unit" is monosubstituted (one oxygen atom shared by the silicon) -O-Si-CH3 I CH• CH• I "D unit" is disubstituted (two oxygen atoms shared by the silicon) -O-Si-O- I CH• O I "T unit" is trisubstituted (three oxygen atoms shared by the silicon) -O-Si-O- I CH• O I "Q unit" is tetrasubstituted (four oxygen atoms shared by the silicon) -O-Si-O- I o If organofunctional groups other than carbon are introduced, the group is given an asterisk (*), which is added to its designation. "M* unit" is monosubstituted (two oxygen atoms shared by the silicon) with organofunctionality CH 3 I -O-Si-CH 3 I R
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)