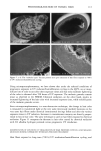
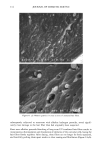
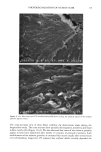
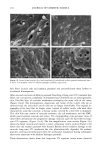
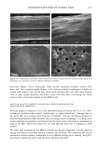
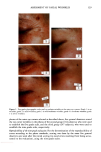
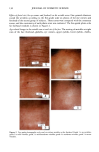
90 JOURNAL OF COSMETIC SCIENCE (9) (10) (11) (12) (13) (14) (15) (16) (17) (18) (5) L. E. Jordana, K. P. Ghiggino, I. H. Leaver, C. G. Barraclough, and P. Cole-Clarke, Depth profile analysis of keratin fibers by FTIR photoacoustic spectroscopy, Appl. Spec., 48, 44-49 (1994). (6) M. Joy and D. M. Lewis, The use of Fourier transform infrared spectroscopy in the study of the surface chemistry of fibers, Int. J. Cosmet. Sci., 13, 249-361 (1991). (7) J. E. Katon, G. E. Pacey, and J. F. O'Keefe, Vibrational molecular microspectroscopy, Am. Chem. Soc., 58, 1-7 (1986). (8) R. G. Messerschmit, "Minimizing Optical Nonlinearities in Infrared Microspectroscopy," in Infrared Micro•pectroscopy: Theory and Applications, R.G. Messerschmit and M.A. Harthcock, Eds. (Marcel Dekker, New York and Basel, 1972), Vol. 6, pp. 1-19. A.J. Soreruer and J. E. Katon, Diffraction-induced stray light in infrared microspectroscopy and its effects on spatial resolution, Appl. Spec., 45, 1633-1640 (1991). G. L. Carr and G. P. Williams, Infrared microscopy with synchrotron radiation, Proceed SPIE Conf, 3153, 125-132 (1997). E. Bramanti, F. Ronca, L. Teodori, M. L. Trucia, F. Papineschi, E. Benedetti, G. Spremolla, P. Ver- gamini, and E. Benedetti, A new approach to the study of human hair by means of FT-IR microscopy, J. Soc. Cosmet. Chem., 43, 285-296 (1992). C. Atsuta, A. Fukumashi, and M. Fukuda, Mechanism of isolation of human hair cuticle with KOH/1-butanol solutions, J. Soc. Cosmet. Chem., 46, 281-290 (1995). K. S. Kalasinsky, J. Magluilo, Jr., and T. Schaefer, Hair analysis by infrared microscopy for drugs of abuse, Forensic Sci. Inc., 63, 253-260 (1993). K. S. Kalasinsky, J. Magluilo, Jr., and T. Schaefer, Study of drug distribution in hair by infrared microscopy visualization, J. Analyt. Toxicol., 18, 337-341 (1994). K. S. Kalasinsky, Drug distribution of human hair by infrared microscopy, Cell. Mol. Biol., 44, 81-88 (1998). G. L. Carr, A. Hanfland, and G. P. Williams, Midinfrared beamline at the National Synchrotron Light Source port U2B, Rev. Sci. Instrum., 66, 1643-1645 (1995). G. L. Carr, A. Hanfland, and G. P. Williams, Performance of an infrared microspectrometer at the NSLS, Rev. Sci. Instrum., 66, 1490-1492 (1995). J-L. Bantignies, G. Fuchs, L. Cart, P. Dumas, and C. Wilhelm, Industrial applications of accelerator- based infrared sources. Analysis using infrared microspectroscopy, Proceed SPIE Conf, 3153, 125-132 (1997). (19) C. R. Robbins, in Chemical and Physical Behavior of Human Hair, 2nd ed. (Springer-Verlag, Piscataway, NJ, 1988), pp. 39-68. (20) C. Zviak and R. P. R. Dawber, "Hair Structure, Function, and Physicochemical Properties," in The Science of Hair Care (Dermatology Series, Vol. 7) (Marcel Dekker, New York and Basel, 1986), pp. 1-30. (21) L. E. Jordana, K.P. Ghiggino, I.H. Leaver, and P. Cole-Clarke, Application of FT-IR step-scan photoacoustic phase modulation methods to keratin fibers, Appl. Spec., 49, 361-366 (1995). (22) L. M. Miller and L. Carr, in preparation.
j. Cosmet. Sci., 51, 91-101 (March/April 2000) Structure-function relationships of dimethicone copolyol NELSON E. PRIETO and ANTHONY J. O'LENICK, Petroj•rm Inc., Fernandina Beach, FL (N.E.P.), and Siltech USA, Dacula, GA (A.J. O'L.). Accepted for publication January 31, 2000. INTRODUCTION Throughout the 1990s dimethicone copolyol (DMC) surfactants and their derivatives have been an important and growing class of surface-active agents. These materials are used in a diverse area of applications such as cosmetics, textiles, coatings, lubricants, and detergents, due to their ability to provide maximum surface-active properties in a cost-effective manner (1-3). Despite the growing use of these silicone-based compounds, studies regarding the basic understanding of the effect of their structure on surfactant properties remain limited, relative to the information openly available for organic sur- factants. The present study was undertaken to determine the effect of DMC structure on its surfactant properties. Properties studied included wetting, cloud point, critical micelie concentration (cmc), foaming, solubility, spreading, emulsification, and ocular irritation. The relationship of the results to application is discussed. DIMETHICONE COPOLYOL CHEMISTRY The term dimethicone copolyol has been adopted by the Cosmetics, Toiletry and Fra- grance Association to describe a class of silicone/polyoxyalkylene derivatives. DMC surfactants are a class of compounds that conform to the following general structure: CH 3 CH 3 CH 3 CH 3 CH3-Si--(-O--Si--)•--(O--Si)b--O--Si--CH 3 CH 3 CH 3 (CH2) 3 CH 3 I O-(CH2CH20)x-(CH2CH(CH3)O)yH The nomenclature was developed to reflect the fact that the molecule is (a) a silicone polymer (dimethicone), (b) a copolymer ("copoly" part), and (c) hydroxyl functional ("o1" 91
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)