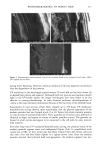
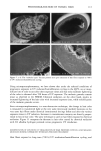
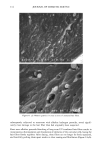
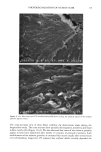
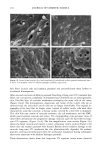
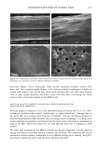
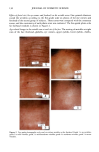
PHOTODEGRADATION OF HUMAN HAIR 105 700 hours at alternating three-hour cycles of humidification (at 95 % RH 40øC) and UV irradiation (50øC RH decreasing from 95% to 10% within 30 min, then remaining at 10% for 2.5 h) and (b) 0, 100, 200, and 300 hours continuous UV irradiation at constant 10% RH and 50øC. At/as Weather-Ometer ©, ("AW"). The "AW" has a spectral distribution ranging from UV through the visible range, simulating sunlight. These exposure conditions correspond to an "average" 45 ø Miami summer sunlight. The energy density at the 340-nm wave- length is kept constant at 0.3 W/m i . This means that at the specific wavelength of 340 nm, the energy density in the QUV (0.97 W/m 2) is approximately three times greater than that of the "AW" (0.3 W/mP). However, in the wavelength range of 300-400 nm, the energy densities of the QUV (300-400 nm yield 5.06 mW/cm 2) and the "AW" (300-400 nm yield 4.46 mW/cm 2) are similar, although the distribution is quite different. The specific spectral distributions in the "AW" in the range of 250-300 nm yield 0.012 mW/cm •, and 400-800 nm yield 36.80 mW/cm 2. The total energy density in the "AW" is 41.272 mW/cm •. Specific exposure conditions of the hair fibers to UV/visible radiation in the "AW" were 0, 100, 200, and 300 hours at constant 20%, 50%, and 70% RH and 50øC. Post-treatment of hair fibers in alkaline hydrogen peroxide. Hair fibers exposed to UV irra- diation/humidification (at 95% RH) cycling were subjected to post-treatment in 6% alkaline hydrogen peroxide from seconds up to two hours. This was done to illustrate and characterize the severe extent of photodegradation inflicted upon the fiber during treat- ment in the QUV. Post-treatment of samples irradiated at low and intermediate humidities with water. UV irra- diation/humidification (at 95% RH) cycling lead to extensive "thinning" of the surface cuticle cells and "fusion" of the scale edges. Differentiation of the surface cuticle cell, so characteristic of the untreated hair fiber, eventually disappears at longer exposure times. We concluded that the presence of moisture in the highly swollen fiber during the humidification cycle is responsible for transporting the degraded, low-molecular-weight protein fragments out of the cuticle cell and possibly into the cortex, thereby causing the collapse and thinning of the surface cuticle cell. Hair fibers irradiated at low humidities (from 10% to 70% RH) in the two fading units do not show this feature of thinning and fusion of the cuticle cell. Apparently, the lack of mobile water in the hair fiber and the lack of swelling of the fiber eliminate the transport of photodegraded fragments out of the cuticle cell. To mobilize the photode- graded proteins, hair fibers irradiated under low RH conditions in both the QUV and "AW" were subjected to a water post-treatment. The fibers were immersed for 60 minutes in lukewarm, deionized water, air-dried, and then examined longitudinally in the SEM. This was to establish whether post-treatment in warm water would result in thinning and fusion of the cuticle cell by diffusing photodegraded materials out of the cuticle cell, assuming that photo-oxidation of the hair proteins had occurred at all. INVESTIGATIVE METHOD Field emission scanning electron microscopy. Longitudinal and cross-sectional segments of untreated and UV-exposed fibers were mounted on double-sided tape and coated with approximately 90 fk of platinum. The hair fiber topography and interior were examined
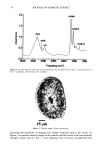
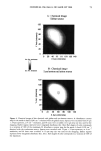
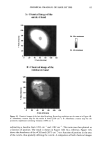
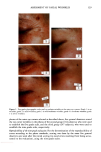
106 JOURNAL OF COSMETIC SCIENCE for photo-oxidative damage in a Hitachi S-4500 digital field emission scanning electron microscope (FESEM). RESULTS AND DISCUSSION BACKGROUND Photochemical degradation of hair affects both hair proteins and hair pigments and has been shown to occur primarily in the wavelength region of 254 to 400. In recent infrared spectroscopic studies of hair exposed to weathering, Dubief (3) shows a decrease in lysine and histidine, and a drastic increase in cysteic acid content in tip ends of hair compared to root ends. This suggests scission of the disulfide crosslinks during the photochemical oxidation process and formation of cysteic acid. This assumption is supported by Rob- bins and Bahl (5), who have shown by electron spectroscopy for chemical analysis (ESCA) that both UV-B (280-320 nm) and UV-A (320-400 nm) oxidize sulfur in hair. Oxi- dation was shown to occur primarily in the fiber periphery, namely, the cuticular sheath, producing a steep gradient to less oxidized hair in the fiber core. These results are not surprising, since disulfide is at its highest concentration in the A-layer and the exocu- ticle, where the highest level of photochemical oxidation occurs. The ESCA spectra suggest that high levels of cystine S-sulfonate and cysteic acid are formed in hair exposed to photochemical oxidation in the wavelength region of 254-500 nm. Our research (6) strongly supports the results of Dubief (3) and Robbins and Bahl (5). Using UV microspectrophotometry, we have shown that high levels of photodegradation products are formed throughout the hair fiber cross section during long-term exposure in the 290-400 nm range. UV irradiation-induced photodegradation products of the hair proteins are revealed by an extension of the absorbance plateau and a shift in peaks from 290 nm in untreated hair to 315 nm in UV-exposed hair. There is also the development of an absorbance shoulder in the 330-340 nm range, well isolated from the absorbance of the bulk of the hair fiber (Figure 1). Formation of the photo-oxidized hair proteins can be traced and mapped, even quantified, by scanning across hair fiber cross sections at the wavelengths of the absorbance shoulder, (}t m -- 330 nm). These photo- degradation products are especially pronounced in blond (unpigmented) Piedmont hair, with the highest level ofphotodegradation occurring in the cuticular region (A-layer and exocuticle), where cystine is at its highest concentration (Figure 2). It is a generally accepted concept that the mechanism for photochemical oxidation of cystine follows the C-S scission pathway, whereby oxidative scission yields S-sulfonic acid that is finally degraded by light to cysteic acid (7,8). In contrast to the chemical oxidation that follows the S-S scission pathway and yields two moles of cysteic acid per mole of reacted disulfide, only one mole of cysteic acid is produced from each mole of reacted disulfide in the C-S scission pathway. The progressive oxidation pathways for the two types of scission are: S-S Scission (chemical oxidation): R-S-S-R --- R-SO-S-R • R-SO2-S-R -• R-SO2-SO-R -• R-SO2-SO2R -• 2R-SO3H C-S Scission (photochemical oxidation): R-S-S-R • R-S-S-OH • R-S-SO2H • R-S-SO3H • R-SO3H + H2SO 4 + R-OH
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)