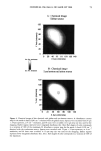
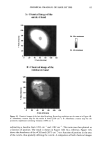
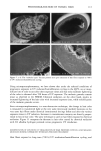
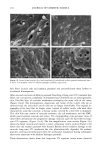
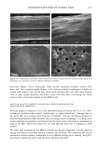
PHOTODEGRADATION OF HUMAN HAIR 111 TRI { - . Figure 5. Representative cross-sectional views of the cuticular sheath of an untreated (al,a2) and a 300-h UV-exposed (b) hair fiber. during severe bleaching. However, chemical oxidation of the hair pigments occurs faster than the degradation of the proteins. UV irradiation in the wavelength region between 254 and 400 nm has been shown (9) to degrade hair protein and pigment. Although both hair proteins and pigments absorb light in the UV/visible region, the longer wavelengths have been found to be less effective in causing photodamage. As with chemical oxidation, photodegradation of cystine is the most extensive phenomenon because of the reactivity of the disulfide bond. Examination of cross sections of hair fibers exposed up to 700 hours UV-irradiation/ humidification cycling showed, quite surprisingly, that the physical appearance of the melanin granules had not changed much, if at all. Figure 8a,b shows melanin granules in cross sections of untreated hair fibers. These granules are of various sizes, spherical or elliptical in shape, and appear to consist of smaller granular entities. The granules are housed in small cavities and appear to be connected to the cell walls by some intercel- lular material. Even after long-term UV-irradiation/humidification cycling, the physical nature of the melanin granules appears intact and undegraded (Figure 9a,b). In unpublished work carried out at TRI, we have shown that hair fibers retained their dark brown color and that only a few had been faded slightly to a lighter brown color. Since the melanin granules retained their physical bulk and appearance, the melanin pigment was pro-
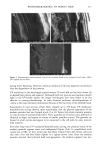
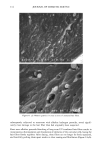
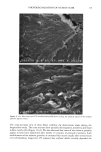
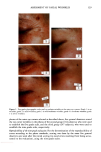
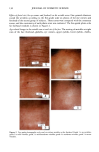
112 JOURNAL OF COSMETIC SCIENCE 30509• 5•0 kV x4.0•k"'ff25•'•& .. Figure 6. a-d: Typical longitudinal views of a long-term UV-exposed hair fiber, displaying severe radial cracking, a typical radiation-induced damage phenomenon. tected from degradation, which, in turn, prevents a loss of hair color. The assumption was made that photochemical oxidation of the melanin chromophor would have to occur in situ if color loss was to occur at all. This is quite in contrast to the swelling and dissolution of the melanin granules by chemical oxidation with hydrogen peroxide. After 1 hour of oxidation with 6% alkaline hydrogen peroxide, moderate disintegration of the melanin granules had occurred (Fig- ure 10b,c). The subgranular structures, which make up the melanin granules, appear more pronounced. There are various stages of degradation, from mile to severe, due to partial solubilization of the melanoprotein. Solubilization appears to occur preferentially from the center of the granules (Figure 10c). After the hair fibers were exposed for four hours to oxidation with 6% alkaline hydrogen peroxide, most of the granular cavities were empty (Figure 10d). The assumption was made that the majority of the cavities were empty because the granules were dissolved away by the peroxide. Progressive solubilization of the melanoproteins is the prerequisite for solubilization and/or dispersion of the melanin pigments. With the melanoprotein dissolved and diffused into the bulk of the hair fiber, the spread-out melanin pigment becomes the unrestricted target of bleaching or loss in color of the human hair. Wolfram and Hall (10) have observed similar behavior with melanin granules isolated from hair and exposed to peroxide bleaching.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)