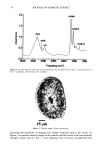
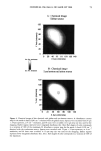
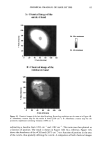
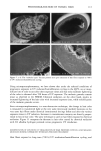
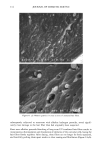
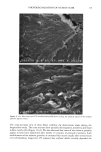
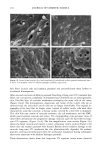
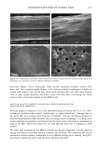
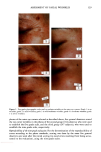
80 JOURNAL OF COSMETIC SCIENCE A' Optical Image 96. x Ax• (•m) X Axis (•n) hat. Abs. maximum : . .. hat. Abs. minimum B: Chemical Image of the cuticle C: Chemical image of the medulla Synchrotron radiation source Synchrotron radiation source Figure 5. Optical and chemical images of hair (synchrotron source). A: Optical image. B: Absorbance contour map of the area of the cuticle for the amide A band (3290 cm-•). Spectra were recorded with 3-pm x 3-pm apertures, at 4 cm -: resolution, and 128 scans were co-added. A 2-prn step size was used for the mapping. Darker regions represent lower integrated absorbance Ont. Abs.). Each degree of color corresponds to a variation of 10% of the maximum. C: Absorbance contour map of the area of the medulla for the amide A band (3290 cm-•). Recording conditions are the same as in Figure 5B. spectrum were 128 scans. The measurement time at each location was approximately one minute. The chemical image of the medulla (Figure 5C) represents the integrated absorbance of the aliphatic CH stretching vibrations centered at 2918 cm -• which are stronger in the medulla, as can be seen from the spectra in Figure 6. This vibration mode is due to asymmetric stretching CH 2 groups that are more intensive in the medulla with respect to the asymmetric stretching modes corresponding to the CH 3 motions centered at 2962 cm-•. The relative intensity of the symmetric modes of the same groups confirm this result. In a previous paper, regarding the infrared fingerprints of the hair fiber as a function of the position along its axis (from the bulb to the shaft), it was shown that the signature of the bulb in the anagen phase in the vCH region is different from that of the shaft (11). The differences observed were comparable to those observed here and suggest
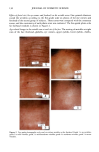
CHEMICAL IMAGING OF HAIR BY IMS 81 0.8 0.7 0.6 0.5 0.4 0.3 0.2 0.1 3050 " • Medulla 2918 cm : ....... Cortex 2962 cm-' ' i i • 2876 crn-' i 2860 cm - , i I I I ß 3000 2950 2900 2850 2800 Frequenc• (cm-') Figure 6. Spectrum of the medulla and cortex. Recording conditions are the same as in Figure 2. that these vibrations are mainly due to the lipids of the cellular membranes. Our results show that the signature of the vCH in the medulla is comparable to those obtained for the bulb in the anagen phase. We can then suggest that the differences are due to an enhancement of the length of the segment of the parraffinic chains of the lipids. We note that our spectra of the medulla are consistent with those obtained for the medulla region by Kalasinsky (15). Imaging the cuticle turns out to be much more challenging and requires identifying a spectroscopic signature indicating its presence. Such a signature can be found by fol- lowing the integrated absorbance of the amide A band (3290 cm -1, -3 pm) as a function of the position in the area of the cuticle, shown in Figure 5B. A linear map across the sharp edge of a polyamide film was performed using the same wavelength and under the same experimental conditions as for the hair sample. This yielded a much sharper transition, suggesting that the broad transition for the hair was intrinsic to the surface (cuticle) region (Figure 7). Furthermore, the spectra in the area of the cuticle exhibit a shift and a broadening of the amide I and amide II bands (Figure 8), which are consistent with their sensitivity to the cuticle/cortex interface as measured by photoacoustic spec- troscopy (5,21). However, these wavelengths, which are comparable to the size of the cuticle, cannot be used for the mapping of this area. In Figure 8, the vC=O band centered at 1730 cm -1 is due to the resin. Due to the fact that the thickness of the cuticle (about 4 microns) is approximately equal to the lateral resolution, it was not possible to quantify heterogeneity throughout the cuticle in our experimental configu- ration. We checked the reproducibility of the above results by mapping ten different hair
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)