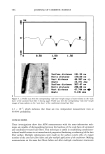
ANTIOXIDANT ACTIVITY OF LICORICE EXTRACT 557 lOO 90 80 70 60 50 40 30 20 lO o o A i i i i i 4 6 8 10 12 Time (Week) B lOO 9o 8o 70 50 30 20 10 0 2 4 6 8 10 12 Time (week) C CB .--•Ext. 2% C CB .--•Ext. 2% .& SM2% ,( BHT2% .& SM2% ,( BHT2% Figure 5. Formulation stability study of 2% w/w hydroquinone cream containing 2.0% extract and commercial antioxidants incubated at 25 ø _+ 0.5øC (A) and 45 ø _+ 0.5øC (B) for three months. mercial antioxidants. According to our procedure, by incorporating hydroquinone after the cream was formed, and together with the solubility property of hydroquinone that is freely soluble in propylene glycol and slightly soluble in oil and water, we had expected that hydroquinone had been absorbed in both the oil and the water phases of the formulation. It is also evident that hydroquinone can be incorporated in the for- mulation in different ways. Besides our procedure, hydroquinone was used in the oil phase with laevo-ascorbic acid as antioxidant, and in the water phase with sodium metabisulfite and ascorbic and citric acids as antioxidants (1). This meant that the types of antioxidants selected for use in the formulation depended on the method of the incorporation of hydroquinone into the system. In our hydroquinone systems containing extract, the licorice acted as both water- and oil-soluble antioxidant agents and dem- onstrated significant protection from oxidative degradation in hydroquinone for three months, in comparison with sodium metabisulfite and BHT that were water-soluble and oil-soluble, respectively. CONCLUSION The licorice extract at 0.5% and 1.0% can be used as a double-action (both water- and oil-soluble) antioxidant, having 76% and 78% (at 25øC) and 55% and 60% (at 45øC) of hydroquinone remaining, respectively, after three months. These results indicate that
558 JOURNAL OF COSMETIC SCIENCE licorice extract is more effective than sodium metabisulfite and BHT, and may be used as a substitute for commercial antioxidants in oxidation-sensitive formulations. REFERENCES (1) A. Manosroi, M. Abe, and J. Manosroi, Comparison of antioxidant activity of extract from seeds of white pepper (Piper nigrum, Linn.) to commercial antioxidants in 2% hydroquinone cream, J. Cosmet. Sci., 50, 221-229 (1999). (2) R. Gr Harry's Cosmeticology (Chemical Publishing Co., New York, 1996). (3) D. Mantle, F. Eddeb, and A. T. Pickering, Comparison of relative antioxidant activities of British medicinal plant species in vitro, J. EthnopharmacoL, 72, 47-51 (2000). (4) T.J. Vanderjagt, R. Ghattas, D.J. Vanderjagt, M. Crossey, and R. H. Glew, Comparison of the total antioxidant content of 30 widely used medicinal plants of New Mexico, Li• Sci., 70, 1035-1040 (2002). (5) C. F. Duffy and R. F. Power, Antioxidant and antimicrobial properties of some Chinese plants, Int. J Antimicrob. Agents, 17, 527-529 (2001). (6) J. Vaya, P. A. Belinky, and M. Aviram, Antioxidant constituents from licorice roots: Isolation, struc- ture elucidation, and antioxidative capacity toward LDL oxidation, Free Radical Biol. Med., 23, 302- 313 (1997). (7) D. Mantle, J. G. Anderton, G. Falkous, M. Barnes, P. Jones, and E. K. Perry, Comparison of methods for determination of total antioxidant status application to analysis of medical plant essential oils, Comparative Biochem. Physiol. B, 121, 385-391 (1998). (8) E. H. Mansour and A. H. Khalil, Evaluation of antioxidant activity of some plant extracts and their application to ground beef patties, Food Chem., 69, 135-141 (2000). (9) J. A. Cook, D. J. Vanderjagt, A. Dasgupta, G. Mounkaila, R. S. Glew, W. Blackwell, and R. H. Glew, Use of the Trolox assay to estimate the antioxidant content of seventeen edible wild plants of Niger, Life Sci., 63, 105-110 (1998). (10) J. A. Vinson and Y. A. Dabbagh, Tea phenols: Antioxidant effectiveness of tea, tea components, tea fractions and their binding with lipoproteins, Nutr. Res., 18, 1067-1075 (1998). (11) P.A. Belinky, M. Aviram, B. Fuhrman, M. RosenNat, and J. Vaya, The antioxidative effects of the isoflavan glabridin on endogenous constituents of LDL during its oxidation, Atherosclerosis, 137, 49-61 (1998). (12) H. Hayashi, N. Hiraoka, Y. Ikeshiro, and H. Yamamoto, Organ specific localization of flavonoids in Glycyrrhiza glabra L., Plant Sd., 116, 233-238 (1996). (13) K. Okada, Y. Tamura, M. Yamamoto, Y. Inoue, R. Takagaki, K. Takahashi, S. Demizu, K. Kajiyama, Y. Hiragy, and T. Kinoshita, Identification of antimicrobial and antioxidant constituents from licorice of Russian and Xinjiang origin, Chem. Pharm. Bull., 37, 2528-2530 (1989). (14) B. Fuhrman, S. Buch, J. Vaya, P. A. Belinky, R. Coleman, T. Hayek, and M. Aviram, Licorice extract and its major polyphenol glabridin protect low-density lipoprotein against lipid peroxidation: In vitro and ex-vivo studies in humans and in atherosclerotic apolipoprotein E-deficient mice, Am. J Clin. Nutr., 66, 267-275 (1997). (15) S. Bemizu, K. Kajiyama, K. Takahashi, Y. Hiraga, S. Yamamoto, Y. Tamura, K. Okada, and T. Kinoshita, Antioxidant and antimicrobial constituents of licorice isolation and structure elucidation of a new benzofuran derivative, Chem. Pharm. Bull., 36, 3474-3479 (1988). (16) M. H. Gordon and J. An, Antioxidant activity of flavonoids isolated from licorice, 43, 1784 (1995). (17) F. S. D'Amelio, Botanicals: A Phytocosmetic Desk Refirence (CRC Press, Boca Raton, FL, 1999). (18) A. Y. Leung and S. Foster, Encyclopedia of Common Natural Ingredients Used in Food, Drags and Cosmetics (Wiley-Interscience, New York, 1996). (19) United State• Pharmacopoeia (USP), XXIV (2000).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)