j. Cosmet. Sci., 54, 559-568 (November/December 2003) Solubilization of sodium cocoyl isethionate JAMES ZIMING SUN, JAMES W. PARR, and MICHAEL C. E. ERICKSON, Advanced Research Laboratories, 151 Kalmus Dr. Suite H-3, Costa Mesa, CA 92626. Accepted for publication July 15, 2003. Presented at the Annual Scientific Seminar of the Society of Cosmetic Chemists, Washington, DC, May 9, 2003. Synopsis Sodium cocoyl isethionate (SCI) has been a predominant ingredient in syndet bar formulation for more than thirty years. Although cost effective and well recognized for good skin compatibility, SCI is not regularly found in liquid detergent systems due to its limited solubility in water. This study focuses on the under- standing of enthalpy of solubilization, equilibrium of solubilization, and the structures and properties of sodium cocoyl isethionate and various surfactants. The purpose of this exercise is to help the formulator to find appropriate surfactant systems to keep sodium cocoyl isethionate in aqueous solution. The solubility of SCI in water is unfavorable in terms of enthalpy of solvation. When setting up equilibrium of solubilization, there are three possible phases, and three methods have been developed to prevent SCI from recrystallizing in aqueous solutions. The first focuses on tying CI ions within micelies made of secondary surfactants. The second focuses on the exchange of sodium ions with ammonium ions (and/or triethanola- monium). The third centers on emulsification of SCI and the subsequent change of micelies into emulsified oil drops. A combination of two or three of these methods will enable the formulator to use SCI as the primary surfactant in liquid detersive systems. INTRODUCTION Owing to its excellent skin compatibility, mildness, and emollient properties, sodium cocoyl isethionate (SCI) has been used extensively in syndet bars (1-6). SCI has become cost efficient because of its broad application in personal care. Chemists try to incor- porate SCI 'within liquid detersive systems in order to take advantage of both its excellent performance characteristics and its cost efficiency. However, SCI is not widely used in liquid cleansing systems because of its limited solubility in water. Some proprietary technologies have been developed to increase the aqueous solubility of SCI (7-9). Zwitterionic detergents (betaines) were discovered to increase the solubility of SCI in liquid detersive compositions (7). In such systems, SCI, betaine(s), and other anionic surfactants (other than SCI) are the three major surfactants. Alkylamphoacetates are another category of surfactants disclosed to increase the solubility of SCI (8). N- methyl cocoyl tautate and laureth sulfate are also used as major surfactants in these proprietary technologies. Nonionic sugar surfactants selected from the group consisting 559
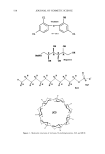
560 JOURNAL OF COSMETIC SCIENCE of alkyl glucose esters, aldobionamides, gluconamides, glyceramides, glyceroglycolipids, polyhydroxy fatty acid amides, and alkyl polyglycosides can also increase the solubility of SCI (9). Selected sugar surfactants, SCI, and free fatty acid are the three major ingredients in most proprietary compositions. These proprietary technologies do not explain why some ingredients dissolve SCI in terms of fundamental physical principles. This article will try to bridge the gap in this understanding by focusing on the enthalpy of solubilization, the equilibrium of solu- bilization, and the structures and properties of SCI and various surfactants. MATERIALS The trade names of materials used in this study are as follows: sodium cocoyl isethionate (85% active): Hostapon SCI 85 © (Clariant GmbH) and Jordapon CI Prill © (BASF) ammonium cocoyl isethionate (25% active): Jordapon ACI-30 G © (BASF) emulsifying wax NF: Polawax © (Croda) disodium laureth sulfosuccinate: Mackanate EL © (Mcintyre) polysorbate 20 and polysorbate 80:Tween-20 © and Tween-80 © (Uniqema). There are many other common surfactants from the Mcintyre Group Ltd, Chemron Corporation, and the Stepan Company. PRINCIPLES Enthalpy of solvation will be used to understand why ammonium cocoyl isethionate (ACI) is more soluble than SCI. Hess's law states that the enthalpy of a reaction is the same whether the reaction takes place in one or several steps (conservation of energy). Born and Haber (10) applied Hess's law to the enthalpy of solubilization of ionic compounds in water (Figure 1). The overall enthalpy of the solubilization process is the sum of two terms: the enthalpy of ion dissociation from the lattice matrix (lattice energy) and the enthalpy of introducing the dissociated ions into the solvent (solvation energy). AHsolubilization = U +AHsolvatio n. Size and other characteristics of ions are two main factors in determining lattice energy and solvation energy. Statistical calculation and experimental analysis show that the enthalpy of solvation is of roughly the same order of magnitude as the enthalpy of lattice energy. Thus the total change of enthalpy of solubilization can be either positive, negative, or zero, depending upon the particular compound. In cases where the enthalpy M-•X'(s) M +(•+ X'(• solubilization •-• M(H20)x+(i) + X(H20)y'(l ) Figure 1. Born-Haber cycle of solubilization of ionic compounds.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)