SOLUBILIZATION OF SCI 561 is negative, zero, or slightly positive, solubilization will take place. If the enthalpy change is too positive, solubilization will not occur. This is to say that ions within the lattice structure will enter solution if the interactions between solute and solvent are strong enough to overcome the lattice energy. The enthalpy of solubilization of SCI is slightly positive in terms of the Born-Haber cycle at room temperature. In another words, the lattice energy of SCI is slightly larger than the combined solvation enthalpy of sodium ion (Na +) and cocoyl isethionate ion (CI-). Sodium cocoyl isethionate has a solubility of about 0.01% by weight at 25øC in water. Alternatively, ammonium cocoyl isethionate (ACI) is very soluble in water at 25øC. A liquid surfactant in the market has 25% active ACI (by weight). However a 25% active liquid solution of ACI is more costly than an 85% active solid of SCI. Thus a substantial use of ACI within shampoos or liquid cleansing formulas would be limited to more expensive "high-end" products. SCI and ACI differ chemically only in their cationic ions, Na + and NH4 +, respectively. Here we focus on the size and characteristics of these cationic ions. Since the ammonium ion is larger in size and has a smaller solvation enthalpy than the sodium ion, -240.1 kJ mol -• for the sodium ion and -132.51 kJ mol -• for the ammonium ion (11), ACI is determined to have smaller lattice energy than SCI because the enthalpy of solubili- zation of ACI should be negative or slightly negative. Smaller lattice energy means that the energy necessary to solvate ACI should be relatively low in comparison to that of SCI. This will become an important point in the discussion of method II. Unfortunately the lattice energy of ACI and SCI, and the solvation enthalpy of CI- ion, are not available in the chemical literature for theoretical calculation. Chemists can find ways to prevent SCI from recrystallizing in aqueous solution at 25øC and below. The following discussion will focus on reaction equilibrium as a means of understanding how to make SCI more soluble in water. When SCI is dissolved in water, the equilibrium reaction is as follows: NaCI(s ) • Na+(1) + CI-o) • Na+(1) + MIC(CI-)(1 ) Le Chatlier's theorem states that if a system in equilibrium is disturbed, it acts to minimize the disturbance. Any change will make the reaction shift to either the right or left in order to reestablish equilibrium. The phase separation model (6) tells us that micelles constitute a new phase, formed in the system at and above the critical micelle concentration. The equilibrium reaction indicates that it is reasonable to assume there are three possible phases involved in the equilibrium: recrystallizing solid (SCI), mo- nometic surfactant ions (Na + and CI ), and micelles (MIC) of pure CI-. Decreasing the concentration of CI-, Na +, or MIC(CI) will shift the reaction equilibrium to the right, which leads to a reduction of solid SCI the solubility of SCI is increased. Increasing the concentration would shift the equilibrium to the left and decrease the solubility of SCI. Krafft temperature is often used to describe the temperature-dependent solubility of surfactants in water. Surfactant solubility will undergo a sharp, discontinuous increase at some characteristic temperature referred to as the Krafft temperature, T x. At tempera- tures below T x, solubility is determined by the crystal lattice energy (U) and the enthalpy change of hydration of the system AHsolvation, as stated in the Born-Haber cycle. At temperatures above Tx, the solubility of the surfactant monomer may increase to the point at which micelle formation begins and aggregate forms become thermo-
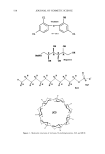
562 JOURNAL OF COSMETIC SCIENCE dynamically stable. In the cases of SCI and ACI, it is apparent that ACI has a lower T (below room temperature), since ACI has a very small lattice energy and SCI has a higher T (close to the boiling temperature of water) due to its large lattice energy. METHODS Three methods of preventing recrystallization of SCI have been developed based on the enthalpy of solubilization and equilibrium. The first focuses on secondary surfactants, which tie up CI- ions within micelies. The second focuses on the exchange of sodium ions with ammonium ions (and/or triethanolamonium). The third centers on introduc- ing emulsifiers and emollients to change micelies into stable emulsified oil drops. METHOD I: SELECTED SECONDARY SURFACTANTS When thinking about SCI solvation, it is important to refer to the equilibrium equation stated above. If one modifies an SCI/water system in such a way as to shift equilibrium to the right, away from solid SCI, then its solubility in water is increased. Modifying pure MIC(CI) with specific secondary surfactants will accomplish this goal both by increasing micelie stability and by increasing the number of aqueous CI- ions that are taken up within micelies. Generally, there are four types of interactions (or molecular interactions) involved in micelie formation (6). They are: (i) hydrophilic/hydrophobic interaction between sur- factant and water molecules (ii) interaction among solvated head groups (generally repulsive) and between the head groups and co-ions (iii) attractive interaction among hydrocarbon tails in separate molecules and (iv) geometric packing constraints derived from the particular molecular structure involved. The last two interactions are very important to the discussion of method I since interactive forces can vary significantly, depending on the surfactants. The second interaction listed will become a focus in method II. In order to tie CI- ions in micelies without recrystallization, one has to choose surfactants that strongly interact with CI- ions. To promote strong interaction with CI- ions, surfactants should have either similar hydrophilic head groups or larger and complicated hydrophilic head groups. The struc- ture of SCI is depicted in Figure 2 along with the structures of other selected surfactants. Anionic surfactants, amphoteric surfactants, and non-ionic surfactants can serve this purpose. In the anionic surfactant category, these are (but not limited to): sodium (or ammonium) dialkyl sulfosuccinates, disodium (or diammonium) alkyl sulfosuccinates, disodium (or diammonium) alkyl ether sulfosuccinates, disodium (or diammonium) alkylamido MEA sulfosuccinates, disodium (or diammonium) alkylamido MIPA sulfo- succinates, disodium (or diammonium) alkylamido PEG-2 sulfosuccinates, sodium (or ammonium) acyl taurates, disodium (or diammonium) acyl glutamates, sodium (or ammonium) acyl lactylates, and sodium acyl sarcosinate. In the amphoteric category (Figure 3) there are alkylamidopropyl hydroxysultaines, sodium (or ammonium) alkylamphoacetates, disodium (or diammonium) alkylampho- diacetates, sodium (or ammonium) alkylamphopropionates, disodium (or diammonium) alkyliminodipropionates, alkylamidopropyl betaines, alkylamidopropylamine oxides, and sodium alkylamphohydroxypropylsulfonate.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)