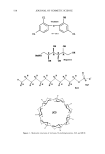
SOLUBILIZATION OF SCI 563 O RC--OCH2CH2SO3Na O NHCH2CH2SO3Na sodium cocoyl isethionate sodium acyl taurate NaOOCH•CH2CH2COONa NH--CR II o disodium acyl glutamate O O OCHC--OCHCOONa CH3 CH 3 sodium acyl lactylate O O RO(CH2CH20)x• CC H2¾H C-- O Na SO3Na sodium alkyl ether sulfosuceinate 0 0 RO•CCH2t•HC--O SO3Na sodium dialkyl sulfosuccinate RO 0 0 CCH2(•HC--ONa SO3Na O NCH2COONa I CH3 disodium alkyl sulfosuccinate sodium acyl sarcosinate Figure 2. Structures of selected anionic surfactants. Non-ionic surfactants require complicated and long-chain polyethylene groups to pre- vent recrystallization of SCI. These surfactants (Figure 4) are polysorbate 20, polysorbate 60, polysorbate 80, alkyl glucosides, PEG-n (n -- 20-80) glyceryl stearates, PEG-n (n = 20-80) glyceryl isostearates, PEG-20-PPG-10 glyceryl stearates, PEG-n (n = 20-80) glyceryl oleates, PEG-n (n = 20-80) glyceryl cocoates, and PEG-n (n: 20-80) glyceryl laurates. Other non-ionic surfactants are less effective in this application. It should be emphasized that micelie stability is an essential element in shifting the SCI solubiliza- tion equilibrium to the right, thus increasing solubility. When a surfactant system consists of head groups that are similar to or larger and more complicated than those of SCI, micelle stability will increase and keep SCI dissolved.
564 JOURNAL OF COSMETIC SCIENCE o CH3 RC--NHCH2CH2CH2N, -- CH2 ,CHCH2-SO 3 - I CH 3 OH O O RC-NHCH2CH21¾C CH2CH2OH alkyl amidopropyl hydroxysulafine sodium alkylamphoacetate o OH II I RC--NHCH2CH21•ICH2CHCH2SO3Na CH2CH2OH O O RC-NHCH2CH21¾CHONa CH2CH2OCH2COONa sodium alkylamphohydroxypropylsulfonate disodium alkylamphodiaeetate O RC-- NHCH2CH21'jiCH2CH2COONa CH2CH2OH O H O -O-- C CH2CH2 I• i+CH2CH2 C-- ONa R sodium alkylamphopropionate sodium alkyl iminodipropionate O CH 3 O !+H 3 RC--NHCH2CH2CH2i CH2CH2COO- R --NHCH2CH2CO-CH3 CH3 alkylamidopropyl betaine alkylamidopropylamine oxide Figure 3. Structures of selected amphoteric surfactants. Additionally, the concentration of CI- within the average micelie decreases as secondary surfactant is added. When micelies in the stated equilibrium consist entirely of CI-, an increase in the concentration of pure CI- micelies will shift the equilibrium to the left, favoring the formation of solid SCI. However, if one disturbs the system such that the concentration of CI- within micelies decreases, i.e., the micelies are no longer pure, then
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)