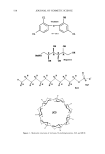
SOLUBILIZATION OF SCI 565 •'•(OCH2CH2)wOH RC-- O(CH2CH20)zCH2iH (OCH2CH2)yOH polysorbate20, R=C 11H23 polysorbate 60, R=C17H35 polycorbate 80, R=C17H33 CH2OH ( H OR OH O (•CH2CH2)xO RC--OCH2-HC•CH2 I (OCH2CH2)yOH alkyl polyglucoside fatty acid monoglyceride ethoxylate Figure 4. Structures of selected non-ionic surfactants. more aqueous CI- will incorporate within these micelies, leading to greater SCI solu- bility. Thus the addition of secondary surfactants not only increases the strength of CI- association with micelies, but also the number of aqueous CI- molecules per micelie increases. By introducing these selected surfactants, one can solubilize over 5-10% SCI in a clear deterslye system. The following two examples illustrate some surfactants in such com- positions. Based on the condition of mildness, the first example (Table I) can be used in a gentle facial cleanser and the second (Table II) is suitable for baby cleansing products. Table I Clear Detersive Formula of Selected Anionic Surfactants With 8% SCI INCI name Trade name Percentage (%) Sodium dioctyl sulfosuccinate Mackanate EL © (30% active) 45.00 Sodium cocoyl isethionate Hostapon SCI 85G © or Jordapon CI Prill © 8.0 Sodium cocoamphoacetate Miranol Ultra C 37 © (37% active) 4.0 Cocamide MEA Areidex CME © 1.5 Thickener q.s. Preservatives q.s. Fragrance q.s. D.I. water To 100%
566 JOURNAL OF COSMETIC SCIENCE Table II Mild Baby Care Detersive Formula of Selected Non-Ionic Surfactants With 7% SCI INCI name Trade name Percentage (%) Polysorbate 20 Tween-20 © i 0.00 Polysorbate 80 Tween-80 © 15.00 Sodium cocoyl isethionate Hostapon SCI 85G © or Jordapon CI Prill © 7 Thickener q.s. Preservatives q.s. Fragrance q.s. D.I. water To 100 It should be noted that as the formulation of cosmetics becomes more sophisticated, chemists will determine the geometric packing of micelies as standard practice, e.g., for spherical micelies, rods, and lameliar structures. It is known that as micelies proceed from spherical to lameliar formations, the density of micelies increases. As the density of micelies increases, the number of free CI- ions decreases. This would push the solubility equilibrium to the right. Thus it is very likely that control of micelie for- mation also affects SCI solubility. METHOD II: ION EXCHANGE Even though some specialty surfactants efficiently solubilize SCI in aqueous solution (method I), they are often more expensive than common surfactants such as sodium lauryl sulfate (SLS) and sodium laureth sulfate (SLES), which do not efficiently solubilize SCI. For example, an aqueous solution of 10% SLS or SLES can only solubilize less than 1% of SCI and remain clear. Making more expensive formulas with specialty surfactants defeats the advantage of using a relatively inexpensive material like SCI. In order to incorporate SCI in affordable detersive systems for the mass market, the ion exchange method was developed through detailed examination of equilibrium (12). The ion exchange method can prevent recrystallization of SCI by changing the total enthalpy of solvation from a positive to a negative value. Ion exchange is accomplished by adding ammonium ions from ALS and ALES, or triethanolamonium from TEALS and TEALES, all of which are significantly cheaper than the aforementioned specialty sur- factants. The presence of free ammonium ions (or triethanolamonium) disrupts the ability of Na + to associate with CI- ions and reform SCI. As was already stated, ACI and/or TEACI compounds have lower lattice energies and are more soluble. It thus becomes possible to keep more CI- ions available to form micelies, which clean surfaces. Having more CI- ions also favors interaction and substantivity to skin and keratin surfaces, allowing the user to experience the emolliency and moisturizing properties associated with this molecule. It should be noted the formulator may choose ACI as a secondary surfactant despite the generation of free CI- ions, which would normally shift equilibrium to the left. This is because the solubility contribution by the ammonium ion dominates the equilibrium the reduction in lattice energy by ammonium outweighs any increase in aqueous CI-. Monoalkyl phosphate (MAP) surfactants will serve the same purpose. MAP surfactants have been studied extensively for their excellent foaming properties, exceptional mild-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)