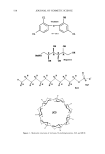
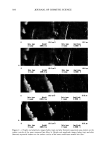
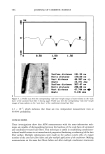
j. Cosmet. Sci., 54, 569-577 (November/December 2003) Nano-structured biphasic polymer film on the hair surface from PEGylated polymer latexes HEE-KYUNG JU, JIN-WOONG KIM, JI-YOUNG PARK, HAK-HEE KANG, SEONG-GEUN OH, and KYUNG-DO SUH, Amore-Pacific ROD Center, 314-1, Bora-ri, Gihe•ng-eup, Yongin-d, Gyeonggi-do 449-726 (H.-K.J., J.-W.K., J.-Y.P., H.-H.K.) and Department of Chemical Engineering, College of Engineering, Hanyang University, Seoul 133-791 (S.-G.O., K.-D.S.), South Korea Accepted for publication July 29, 2003. Synopsis In this study, biphasic polymer latexes were synthesized by surfactant-free-emulsion polymerization of butyl methacrylate, poly(ethylene glycol) methyl ether methacrylate, and 2-(methacryloyloxy) ethyl trimethyl ammonium chloride. The latexes synthesized were composed of hydrophobic core phase and hydrophilic shell phase. Nano-structured film morphology could be obtained by annealing the biphasic polymer latexes between the two transition temperatures. It was found that the unique film morphology gave a viscoelastic property to the film. Scanning electron microscope and atomic force microscope images revealed that the biphasic polymer latexes deposited effectively onto the entire hair surface upon conditioning with 1 wt% polymer concentration in water. Consequently, they formed a smooth polymer membrane thereon, showing a high potential for a new hair cosmetic ingredient. INTRODUCTION Hair styling has been emphasized as a useful tool to express beauty. It has been required that the styling of hair should be performed conveniently, without causing any damage to the hair. Until recently, many different formulations, including hair-setting lotion, hair gel, hairspray, and blowing agents, have been developed and used widely. Usually, the incorporation of polymers into the formulation is an area of particular interest for the achievement of intended physical properties in hair cosmetics. This has a big advantage in that we can easily get good holding power, prolonged curl retention, low stickiness, lack of flaking, gloss, transparency, a good feel, etc. (1-3). However, most of the polymers are dissolved in organic solvents, which causes an undesirable odor and incompatibility with other additives. To solve this problem, the incorporation of poly- mers with a form of latex has been proposed. Actually, Yahagi and Suzuki reported that Address all correspondence to Jin-Woong Kim. 569
570 JOURNAL OF COSMETIC SCIENCE a good styling effect on hair could be induced using submicron polymer particles (4,5). Also, according to some patents, polymer latexes having a low transition temperature deposit well onto the hair surface, finally forming a soft polymer film (6,7). In this contribution, we introduce a useful hair treatment system in which biphasic polymer latexes are used to form a nano-structured film membrane on the hair surface. The biphasic polymer latexes are synthesized by controlling the compatibility between hydrophobic components and hydrophilic components in the polymer molecule (8,9). In order to exclude the effect of surfactants, the latexes are prepared by surfactant-free emulsion polymerization. This paper then investigates the effectiveness of the biphasic polymer latex system on the film property and the deposition efficiency on the hair surface. EXPERIMENTAL MATERIALS Butyl methacrylate (BMA), poly(ethylene glycol) methyl ether methacrylate (PEG-MA, Mw = 300, l100, 2000 g' mol-•), 2-(methacryloyloxy) ethyl trimethyl ammonium chloride (METAC), and 2,2'-azobis(2-methyl propionamidine) dihydrochloride (V-50) were purchased from Aldrich Chemical Co. The inhibitor in the monomers was removed using a removing column (Aldrich). Distilled deionized (DDI) water was used through- out the process. SYNTHESIS OF BIPHASIC POLYMER LATEXES The latexes were synthesized by surfactant-free emulsion polymerization. First, V-50 (0.2 g) and DDI water (360 g) were weighed into a four-necked round flask equipped with reflux condenser, nitrogen inlet apparatus, and mechanical stirrer. Under a nitrogen atmosphere, the reactor was submerged in a thermosrated water bath and stirred with a rotation speed of 300 rpm at 75øC. After 10 min, the monomer mixture (40 g) com- posed of BMA, PEG-MA, and METAC was added to the reactor and polymerized for 10 h. The composition and designation of each sample are listed in Table I. Table I Polymerization Composition and Size Characteristics 'L Composition (wt%) Avg. Dn (nm) Symbol b BMA PEG-MA METAC DLS TEM PBMA 100 -- -- 505 453 NAS300 80 20 -- 475 386 NASl100 80 20 -- 450 347 NAS2000 80 20 -- 330 298 NAS300-M5 75 20 5 287 244 a 0.5 wt% V-50 as an initiator based on total monomer weight 10 wt% solid content. b PBMA is the abbreviation ofpoly(butyl methacrylate). In the symbolization ofNASo•-M[3, o• and [3 mean the molecular weight of PEG in PEG-MA and the concentration of METAC, respectively.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)