POLYMER LATEXES ON THE HAIR SURFACE 571 LATEX CHARACTERIZATIONS The particle size of the latexes synthesized was measured by dynamic light scattering (DLS, Zetasizer 3000HS, Malvern, UK). Particle morphology was observed by a trans- mission electron microscope (TEM, JEOL 2010). Differential scanning calorimetry (DSC, TA Instruments, DSC 910) was used to determine the transition temperature. DSC traces were recorded from -100øC to 100øC, with a heating rate of 5øC/min under $ 2 flow. The mechanical properties of latexes were determined using an Instron uni- versal tensile testing machine at room temperature. In the DSC and Instron measure- ments, the latex film was prepared by annealing the latexes at 50øC for two days in a Teflon square molder. HAIR COATING PROCESS The applicability of the biphasic polymer latexes to hair cosmetics was evaluated by observing their degree of deposition on the hair surface. A bundle of human hairs was impregnated in the diluted latex dispersion (1 wt% polymer content, pH 6.5). After being treated for 1 min, the hairs were dried fully at room temperature. In the case of thermal treatment, the dried hairs were annealed at 80øC for 10 min. The surface characteristics of the treated hairs were then observed by a scanning electron microscope (SEM, Hitachi S-4300). The more exact topography of the treated hairs was observed again with an atomic force microscope (AFM). In the AFM measurement, a latex-treated hair was adhered onto freshly cleaved mica. The sample was observed in air by using a commercial AFM (AutoProbe LS, PSI-LS) equipped with microfabricated V-shaped silicone nitride cantilevers (force constant: 0.05 mN/m) on a scanner using the contact mode. RESULTS AND DISCUSSION CHARACTERISTICS OF BIPHASIC POLYMER LATEXES After surfactant-free emulsion polymerization, the biphasic polymer latexes obtained have a molecular structure composed of BMA, PEG-MA, and METAC units (Scheme 1). Figure 1 shows TEM images of the polymer latexes with the molecular weight of PEG in the absence of METAC. As the molecular weight of PEG increases, the latex size decreases gradually. This happens because the surface of the latexes is richly covered with PEG chains, which are responsible for the enhanced dispersion stability (8,9). Moreover, the surface activity is enhanced as a function of the length of the PEG chains (10). In ?H3 ?H3 )y C=O C=O I I o o I (CH2)3CH3 CH3 (CH2"C)z C=O I o (•H 2 CH 20)nCH 3 I•H 2 CH 2 N(CH3)3CI Scheme 1.
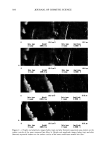
572 JOURNAL OF COSMETIC SCIENCE .•Onm • 50Ohm Figure 1. TEM images of biphasic polymer latexes: (a) PBMA. (b) NAS300. (c) NAS! 100. (d) NAS2000. DLS measurement, even though the effect of the PEG chains shows a similar trend, the latex sizes are somewhat larger than those determined by TEM measurement. This result is related to the effect of hydrodynamic volume. In the aqueous dispersion, the PEG- ylated latexes have a larger hydrodynamic volume, compared with non-treated latexes. In order to examine the effect of a cationic monomer on the latex formation, the emulsion polymerization was carried out in the presence of 5 wt% of METAC. The latex size was remarkably reduced by incorporating the cationic group into the polymer backbone. This reveals that the cationic group makes a big contribution in improving the electrostatic stability of the latex particles (11). The composition and size charac- teristics measured are summarized in Table I. FILM PROPERTIES OF BIPHASIC LATEXES In order to examine the film property, the biphasic latexes were cast in a Teflon molder. The film prepared with the latexes containing 2000 g ß tool • of PEG was very sticky due to the high molecular weight, resulting in a very soft continuous film phase. Therefore, considering basically the film properties, it was established that the molecular weight of PEG should be located between 300 and 1100 g ß tool •. In this consider- ation, the molecular weight of PEG was selected at 300 g ß mol -•. Figure 2 shows the DSC thermograms of the biphasic latex films. PBMA and NAS300 films show only one transition temperature, at around 26øC. On the other hand, NAS300-M5 shows two transition temperatures at -57.5øC and 25.5øC, respectively. The results indicate that
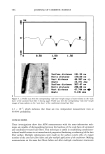
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)