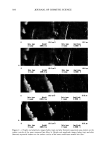
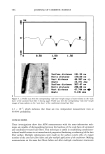
POLYMER LATEXES ON THE HAIR SURFACE 573 E 57.5øC • 25.5oc 26.5øC 26øC -------_ t ß ß i ß -100 -50 0 50 100 Temperature (øC) Figure 2. DSC thermograms of biphasic polymer latex films: (a) PBMA. (b) NAS300. (c) NAS300-M5. the phase separation between PBMA and PEG was facilitated by the introduction of the cationic group into the polymer backbone. It has been known that in the polymer latex system, a high transition temperature contributes to the formation of films on the hair surface, while a low transition temperature exhibits a good deposition profile (7). It is then possible to say that the biphasic latex system having two transition temperatures can achieve the two functions simultaneously. Figure 3 shows the mechanical property of biphasic polymer latex films. In the stress- strain curves, PBMA latex film shows a typical rigid polymer pattern. However, by adding PEG into the polymer composition, the tensile elongation increases on the other hand, the tensile strength decreases steeply. That is, the property of PBMA latex film changes from brittleness to flexibility with the aid of soft PEG moiety. The incorpo- ration of cationic groups slightly lowers tensile elongation, but the stiff property of PBMA latex film can be modified satisfactorily. In the analysis of mechanical property, it is evident that the biphasic latex film has viscoelasticity. APPLICABILITY TO HAIR COSMETICS To evaluate the ability to form a film on the surface of human hair, the biphasic latexes were deposited on the hair and the texture was examined with SEM. Figure 4 shows SEM images of latex films on the hair surface. It is clear that that the biphasic latexes readily covered the hair, resulting in a fine membrane on the hair surface. The coating ability was dependent on the functional group in the latexes. Especially, the cationic group played a crucial role in determining the degree of deposition of latexes on the hair surface. In the absence of the cationic group (NAS300), the deposition of biphasic latexes progressed partially (Figure 4b). However, in the presence of the cationic group (NAS300-
574 JOURNAL OF COSMETIC SCIENCE 5.0 4.0 3.0 2.0 1.0 0.0 0 50 100 150 200 250 Strain (%) Figure 3. Stress-strain curves of biphasic polymer latex films: (a) PBMA. (b) NAS. (c) NAS-M5. M5), the biphasic latex coated the entire hair (Figure 4c), which is attributed to the ion-ion interaction between hairs and positively charged latexes. The topology of latex films on the hair surface was examined by AFM. Figure 5 shows the AFM images of the hair surface. A naked hair exhibits a rough surface because of the cuticles located in the exterior layer. However, after the hair was coated with NAS300- M5, it could be observed that the latexes deposited evenly on the hair surface and formed a film topology corresponding to a spherical cap structure. In addition, even after the film was annealed thermally at a high temperature, the spherical cap structure could be observed in the AFM images (Figure 5c). This film morphology was attributed to the limited interdiffusion between polymer chains, which was induced by the distinctly phase-separated latex structure. In general, the latex film is formed in three steps: water evaporation, particle deformation, and interdiffusion between particles (12,13). This process is largely influenced by several factors, such as the transition temperature of polymers, water resistance, and the surfactants (14-18). In addition to those factors, the compatibility between polymers also plays an important role in determining the final morphology of films (19). In our study, the biphasic polymer latex is composed of hydrophobic PBMA phase in the core and hydrophilic PEG and the cationic group in the shell. With reference to the topology shown in Figure 5, it can be said that PEG-rich shells were mixed selectively by the chain migration between adjacent particles and that the phase mixing between the hydrophobic PBMA core and the PEG-rich shell did not occur even at the high annealing temperature. CONCLUSIONS In this study, the biphasic polymer latexes composed of PBMA in the core and the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)