590 JOURNAL OF COSMETIC SCIENCE Antioxidants have recently been added to formulations to deactivate ROS levels (12) however, similar to our limited understanding of sunscreen-mediated ROS photopro- tection, whether or not the addition of antioxidants enhances photoprotection against UV-induced ROS generation is not well understood. This can be attributed predomi- nantly to limitations in technology that, until recently, have not allowed for the study of cellular processes within the opaque and heterogeneous environment of the skin. Clearly, understanding the effect of sunscreens and antioxidants upon the level of UV- induced ROS generated under biologically relevant conditions (i.e., in viable human skin, at a commonly obtained UV dose) will aid our understanding of the efficacy of these formulations and lead to improved photoprotection. We have recently developed a two-photon fluorescence microscopy method with sub- micron spatial resolution and submillimeter depth penetration in live human ex viva skin to determine UV-induced ROS levels (1). Following UV irradiation, at a dose equivalent to two hours of North American summer solar UV (1600 J m-2), of human breast tissue, ca. 10 -4 moles of •O 2, H202 and/or ONOO- were generated within each epidermal stratum (1). As discussed herein, two-photon fluorescence microscopy serves as an excellent tool to determine the photoprotective effects of sunscreens and antioxidants upon ROS levels generated in epidermis irradiated by a commonly obtained UV dose (1600 J m-2). MATERIALS AND METHODS MATERIALS Reagents. N,N' -[[3 ',6' -bis(acetyloxy)- 3-oxospiro[isobenzofuran- 1 (3 H),9' -[9H]xanthene]- 2••7•-diy•]bis(methy•ene)].bis•N.[2.[(acety••xy)meth•xy]-2.•x•ethy•]].bis[(acety••xy)- methyl] (calcein-AM), dihydrorhodamine-123 (DHR) and fluorescein diphosphate (FDP) were obtained from Molecular Probes. Sunscreen/antioxidant combinations were provided by Roche Vitamins Inc. Table I lists the active ingredients studied. Two sunscreen formulations with different sun protection factors (SPF) were tested. All ingredients are listed by %-weight. The SPF 8 sunscreen contained 4% octylmethoxy- cinnamate (OMC, Parsol © MCX) and 2% avobenzone (Parsol © 1789). The SPF 15 sunscreen contained 7.5% OMC and 3% Parsol 1789. Vitamin E acetate and sodium ascorbyl phosphate were added to the formulations at 2.5 %. Skin samples. Ex viva human skin (breast and facial) was obtained following patient- requested surgery. Samples were obtained with approval from the University of Illinois Internal Review Board. Skin from two individuals was studied. Both skin samples were Table I Active Ingredients Listed by %-Weight SPF 8 SPF 15 Ingredient -E/C +E +E/C -E/C +E +E/C Octylmethoxycinnamate (Parsol © MCX) Avobenzone (Parsol © 1789) Vitamin E acetate Sodium ascorbyl phosphate (STAY-C © 50) 4 4 4 7.5 7.5 7.5 2 2 2 3 3 3 0 2.5 2.5 0 2.5 2.5 0 0 2.5 0 0 2.5
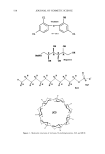
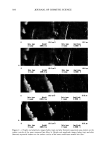
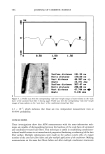
ANTIOXIDANT PHOTOPROTECTION 5 91 lightly pigmented, although, because of their ex vivo nature, their types were not classified. Sample 1 was obtained from the breast of one individual sample 2 was facial skin from a different individual. Age and gender are not specified due to the require- ments of the University of Illinois Internal Review Board. Samples were acquired immediately post-surgery and stored at 4øC in indicator-free RPMI media (Life Tech- nologies) supplemented with gentamicin and L-glutamine until use. Photoirradiation equipment. UV irradiation of each skin sample was achieved using two U¾ fluorescence bulbs (T120W/12RS, Philips). The lamp source peaked near 310 nm in the UVB before tapering into the UVA, where 20% of the photons came between 320 nm and 400 nm. Thus, the data acquired are predominately due to UYB-induced reactions. UV irradiance is determined using an energy meter (Model 1825-C, New- port). A total irradiance of 1600 J m -2 was selected for two reasons. First, the amount of DHR is not fully converted to rhodamine-123 at this irradiance, which indicates that the amount of DHR present is sufficient to react with the quantity of ROS by a dose of 1600 J m -2 (l). Second, an irradiance of 1600 J m -2 is a biologically significant U¾ dose and can be obtained from two hours of noonday summer solar UV exposure in the northern hemisphere (13). TWO-PHOTON FLUORESCENCE IMAGING MICROSCOPY AND ROS DETECTION IN SKIN Two-photon excitation is achieved when a molecule simultaneously absorbs two photons at the excitation wavelength. Typically, two-photon excitation is achieved using an ultrafast, pulsed, near-IR laser system (785 nm, 10 -•5 s, titanium:sapphire). Many are familiar with confocal microscopy, which is commercially available. However, compared to UV or visible confocal one-photon sources, two-photon excitation is advantageous for sectioned imaging of the skin, providing reduced photobleaching of the probe fiuoro- phore and limited photodamage to the sample. In addition, Masters et al. found that the absence of a pinhole in two-photon excitation allows for greater depth penetration into skin compared to UV confocal excitation (14). As a result, two-photon excitation pro- vides submicron spatial resolution with submillimeter depth penetration such that data can be acquired within the cells of each epidermal stratum and through to the upper derreal layers. In two-photon fluorescence intensity imaging microscopy, the sample is incubated with a fluorophore that can be excited at the two-photon excitation wave- length. Fluorescence probes for a number of chemical reactions or properties within biological samples are available, including those for ROS detection and pH within skin, where a change in fluorescence intensity or lifetime relative to a control sample yields information on the sample (1,15,16). For example, to study ROS photoprotection within human skin, a five-step procedure is followed to determine the effect of a topical formulation, like a sunscreen- or antioxi- dant-containing crbme, upon UV-induced ROS levels within ex vivo skin samples. First, approximately 2 mg cm -2 of the formulation is applied to the surface of a skin sample (-0.5 cmx 0.5 cm) and incubated for three hours at 4øC to maintain tissue viability. Second, the sample is incubated for ten minutes at room temperature in a solution containing the ROS-detecting probe dihydrorhodamine (DHR, 100 •M in 2:1 PBS/ EtOH). DHR is nonfluorescent until it reacts with •O2, H202, and/or ONOO- (and potentially other ROS), forming fluorescent rhodamine- 123 (R 123, emission maximum 525 nm) (Figure 1A). Third, the sample is imaged to obtain background (before UV) fluorescence levels for each epidermal stratum. Image areas are between 625 •m 2 and
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)