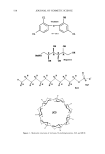
592 JOURNAL OF COSMETIC SCIENCE A non-fluorescent dihydrorhodam•e ROS •N (••NH•+ COOCH• fluorescent rhodamine-123 o o o II tl , •(CI•COCH•OCCH• (CH•OC•OCCH,• OCOCH•. i.•'.O. • .OCOCH• non-fluorescent calcein-am Esterase (COOH•)•NCH• CH•N(COOH0• HO i•o fluorescent calcein Figure 1. Molecular probes used to detect ROS (A) and esterase activity (B). 4000 pm 2 and are acquired ca. every 10 pm, beginning at the stratum corneum surface. Images in the z-direction as little as 2 pm apart can be acquired if desired. Fourth, the skin sample is irradiated by UV (T120W/12RS, Philips), and finally re-imaged. Each image acquired is composed of 256 x 256 pixels. A discussion of the UV source and its differences relative to solar UV radiation is out of the scope of this paper the reader is referred to other work (1). In our experiments two-photon excitation is achieved by a Nd:YVO 4 pumped (Millenia, Spectra-Physics) titanium:sapphire laser (Tsunami, Spectra-Physics), whose fundamental at 785 nm is coupled through the epifiuorescence port of a Zeiss Axiovert microscope. Fluorescence from the samples is collected by a photomultiplier tube (R3996, Hama- matsu). IMAGE ANALYSIS To determine the reduction in UV-induced ROS at depth z due to a test formulation, equation 1 is used: % - Reduction(z) = 100 - 100 (I(z) .... p/e• (1) • I(Z)control / At each epidermal depth z, the fluorescence intensity is calculated over the entire image. These intensity data are averaged together for each skin area studied (I(z)s•,e/). At least two unique areas are imaged per skin sample. I(Z)co,,ro • is calculated identically for each area, where the control images are those acquired on skin incubated with DHR only and indicate the control level of ROS that is generated at the UV irradiance used (1600 J m-2). The average reduction in ROS (% reduction•v a) is calculated by averaging all % reduction(z) values calculated from each tissue sample.
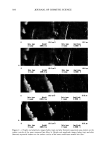
ANTIOXIDANT PHOTOPROTECTION 593 ESTERASE AND PHOSPHATASE ACTIVITY To facilitate penetration of the enzymatic activity dyes, the stratum corneum of each skin sample (-0.5 cm x 0.5 m) was removed by tape stripping (Scotch clear tape). These were the only samples in which the stratum corneum was removed. Without removal of the stratum comeurn, the enzymatic-activity dyes did not penetrate and enzymatic activity could not be detected. Each sample was incubated at room temperature in a 50-1aM calcein-am or fluorescein diphosphate solution (2:1 PBS:EtOH) for ten minutes. Cal- cein-am is nonfluorescent until active esterases act upon it to form fluorescent calcein (Figure lB, emission maximum 520 nm). Similarly, fluorescein diphosphate is nonflu- orescent until phosphatase activity leads to the formation of fluorescent fluorescein (emission maximum 514 nm). The samples were imaged on the two-photon microscope. Data from two control samples were also acquired on the two-photon microscope: the autofluorescence of the skin sample was collected and the fluorescence intensity of the calcein-am and FDP solutions used for incubation was also collected. The data were used for comparison with the images of skin incubated with dye to determine if enzymatic activity took place. RESULTS AND DISCUSSION Because their photoreactions may have a dramatic impact upon photoaging and photo- carcinogenesis, we focus upon ROS generation in the keratinocytes of the strata granu- losum, spinosum, and basale. Following irradiation by 1600 J m -2 UV of the ex vivo breast tissue tested, a dramatic increase in rhodamine-123 fluorescence, and thus ROS levels, was detected in all viable epidermal strata (Figure 2A-C) and in the collagen-rich dermis (Figure 2D). The images indicate the fluorescence intensity expected in the different strata following a UV dose of 1600 J m-2 they represent our controls for the two skin samples. As the images show, ROS are generated predominantly in the cytoplasm of the keratinocytes. This can be seen by noting the red and yellow areas surrounding the blue nuclei. The blue colors indicate the absence of detectable ROS. Because our ROS probe may not have penetrated the nuclear membrane, we cannot comment upon nuclear ROS generation at this time. These images are consistent with previous data (1) and result from the presence of UV-absorbing chromophores present in the cytoplasm of the cells (NADH/NADPH, 5 200 5 200 5 200 5 100 Figure 2. Two-photon fluorescence intensity images of R123 emission in viable ex vivo human breast skin following irradiation by 1600 J m 2 UV. These images represent the control data used to calculate the effect of the formulations tested upon ROS levels. The corresponding intensity scale bar is displayed below each image, where red represents the maximum number of ROS generated following irradiation. Blue indicates the absence of ROS. Images are displayed of the strata granulosum (A), spinosum (B), basale epidermis (C), and dermis (D). Each image is 50 t•m x 50 t•m.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)