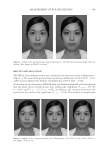
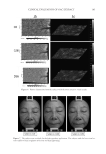
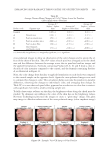
JOURNAL OF COSMETIC SCIENCE 196 without effect pigments on each side of the face over the course of fi ve days. Clinical grad- ing, colorimetric measurements, and digital images were obtained 10 min after applica- tion of the face cream on each of the 5 days. In vivo face cream application. On evaluation days, each panelist was instructed to wash their face 30 min prior to the visit, and not to apply any makeup or personal care prod- ucts. After a check in with the testing center and evaluation to ensure the instructions were correctly followed, the panelists were acclimated for a minimum of 30 min. After acclimation, the panelists were provided ~0.5g of two test face creams, one being the control cream and the second being a randomly selected test cream A–E that is formu- lated with a different effect pigment. The panelists were then instructed to apply both creams evenly to different sides of the face and eye area under guidance of a technician to ensure proper application. Both creams were allowed to dry for 10 min prior to any evaluation. Each panelist repeated this process for the 5 days, so that all fi ve different test creams containing products A–E were applied at least once alongside the control cream. Formulations. A conventional face cream (O/W system) was prepared in a total of six dif- ferent versions. The fi rst batch was formulated with no effect pigment added, while the next fi ve batches were prepared with a baseline of 2.5 wt. % of each effect pigment, with weight corrections made as previously explained in the uniform coverage analysis to en- sure even coverage. Since the solvent content in the lacquer used in the coverage analysis is similar to the water content in the formulation, the fi lm created by the formula and the lacquer will have close pigment to dry matrix weight ratios. (Table II) Materials. glyceryl stearate (and) ceteareth-20 (and) ceteareth-12 (and) cetearyl alcohol (and) cetyl palmitate (Emulgade® SE-PF), behenyl alcohol (Lanette® 22), ethylhexyl pal- mitate (Cegesoft® C24), hexyldecanol (and) hexyldecyl laurate (Cetiol® PGL), dicaprylyl ether (Cetiol® OE), sodium polyacrylate (Cosmedia® SP), sodium stearoyl glutamate (Eu- mulgin® SG), Propylene glycol (and) phenoxyethanol (and) chlorphenesin (and) methyl- paraben (Elestab® 388) were obtained from BASF Corporation (Florham Park, NJ). Dimethicone (Xiameter® PMX-200 Sil Fluid 50) was obtained from Dow Corning (Mid- land, MI). Glycerin was obtained from Jeen International Corporation (Fairfi eld, NJ). Xanthan gum (Keltrol® CGT) was obtained from CP Kelco (Atlanta, GA). Pigments used. Mica (Mearlmica® SV), bismuth oxychloride (Pearl-Glo® UVR), mica (and) titanium dioxide (Flamenco® Velvet 120V), Synthetic Fluorphlogopite (and) Titanium Dioxide (Chione™ HD Infi nite White S130V), Calcium Sodium Borosilicate (and) Tita- nium Dioxide (Refl ecks™ Dimensions Luminous White G130M) were obtained from BASF Corporation. Colorimeter measurement and digital images. On each evaluation day, panelists fi rst accli- mated to controlled environmental conditions for at least 30 min. After acclimation, Visia CR (Canfi eld Scientifi c, Inc., Fairfi eld, NJ) digital photos of the panelist’s faces were taken and CR-400 Chroma Meter (Konica Minolta Sensing Americas, Inc., Ramsey, NJ) measurements were obtained on each side of the panelist’s cheeks. After 10 min of test product application, VISIA CR digital photos and Chroma Meter measurements were obtained again. The VISIA CR captures fi ve images at a time, the fi rst being a standard photo with directional lighting, and the second being a diffuse light source. The third captured image is a cross polarized light source, which is to eliminate gloss effect and specular refl ections to observe only the skin coloration. The fourth is a parallel polarized
ENHANCING SKIN RADIANCE THROUGH THE USE OF EFFECT PIGMENTS 197 Table II Formulation of Conventional Face Cream–Control Weight % Phase INCI Control cream Test cream A Test cream B Test cream C Test cream D Test cream E I Glyceryl stearate (and) ceteareth-20 (and) ceteareth-12 (and) cetearyl alcohol (and) cetyl palmitate 5.00 5.00 5.00 5.00 5.00 5.00 Behenyl alcohol 2.00 2.00 2.00 2.00 2.00 2.00 Ethylhexyl palmitate 5.00 5.00 5.00 5.00 5.00 5.00 Hexyldecanol (and) hexyldecyl laurate 3.00 3.00 3.00 3.00 3.00 3.00 Dicaprylyl ether 3.00 3.00 3.00 3.00 3.00 3.00 Sodium polyacrylate 0.80 0.80 0.80 0.80 0.80 0.80 Dimethicone 1.00 1.00 1.00 1.00 1.00 1.00 II Water 74.10 71.60 71.60 71.60 69.10 69.10 Propylene glycol (and) phenoxyethanol (and) chlorphenesin (and) methylparaben 2.50 2.50 2.50 2.50 2.50 2.50 Glycerin 3.00 3.00 3.00 3.00 3.00 3.00 Sodium stearoyl glutamate 0.40 0.40 0.40 0.40 0.40 0.40 Xanthan gum 0.20 0.20 0.20 0.20 0.20 0.20 Mica – 2.50 – – – – Mica (and) titanium dioxide – – 2.50 – – – Synthetic fl uorphlogopite (and) titanium dioxide – – – 2.50 – – Calcium sodium borosilicate (and) titanium dioxide – – – – 5.00 – Bismuth oxychloride – – – – – 5.00 Total % 100.0 100.0 100.0 100.0 100.0 100.0 light source that is used to enhance specular refl ections and observe the lightness proper- ties of the skin surface. The fi fth image is captured under an ultraviolet light source. RESULTS COLORIMETER RESULTS The CR-400 Chroma Meter allows for color measurements directly on skin, by the use of a diffuse light source that shines on the skin and a detector at 0º (or normal to the surface).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)