JOURNAL OF COSMETIC SCIENCE 168 quality assurance standards. Suncare actives (including OMC) are usually analyzed by techniques such as high-performance liquid chromatography (HPLC) (5–8), which often requires high levels of technical expertise, time (method development), and investment in advanced equipment, resulting in high cost per sample analyzed. Therefore, develop- ment of a simple, rapid, precise, accurate, and inexpensive technique which is practically feasible (5,9) would be of profound benefi t to the pharmaceutical industry and preferred to the currently used routine practices in product formulation (10). Accordingly, this report highlights a simple, cost-effective method based on UV-spectrophotometry for the analysis of OMC in sunscreen products. The method has been validated in accordance with the International Council for Harmonisation (ICH) and Association of Offi cial Ana- lytical Chemists (AOAC) guidelines (11,12), with validated repeatability in three types of dosage forms: oil in water (O/W) and water in oil (W/O) emulsions, and mineral oil. The results indicate that this method should be widely applicable, and feasible for incor- poration into suncare product testing protocols. MATERIALS AND METHODS Those cosmetic formulations were of cosmetic grade, whereas those of analytical practices were of analytical grade. MATERIALS AND INSTRUMENTS OMC (BASF, Duesseldorf, Germany) concentrated 1%, 4%, and 7.5% in sunscreen emul- sion (O/W) consisted of isopropyl myristate (Kik.K Oleo, Selangor, Malaysia), isopropa- nol (Top solvent, Bangkok, Thailand), stearic acid (NOF, New York, NY), cetyl alcohol (Godrej industries, Mumbai, India), triethanolamine (Hangzhou, Zhejiang, China), car- bopol 934 (Lubrizol, Wickliffe, Ohio), and deionized water. Oil phases were melted and mixed at 70°C before OMC addition, followed by the addition of water phase containing carbo- pol and triethanolamine to give homogeneous cream. W/O emulsion was formulated with petrolatum (Paraffi n oil, India), emulsifying wax (Anglo-chemical, Free State, South Africa), calcium stearate (Peter Greven, Bad Mü nstereifel, Germany), caprylic/capric tri- glyceride (BASF), jojoba oil (Desert whale, Tucson, AZ), and microcrystalline wax (Hong Huat, Bangkok, Thailand) that were melted together at 80°C, followed by additions of deionized water and OMC. In addition, OMC was separately dispersed in mineral oil (Kesoon Fine Chemical, Maoming, China). The proportions of each ingredient of the prepared formulations were shown in Table I. A UV-visible spectrometer (UV mini 1240 Shimadzu, Tokyo, Japan) was used to record the absorbance. The product was sun protec- tion factor (SPF) monitored by SPF analyzer (Optometrics LLC/SPF-290F, Miami, FL 13). VALIDATION OF OMC ANALYSIS METHOD Specifi city. OMC (0.05 g) was weighted (BP 2215 Sartorious, Göttingen, Germany) into a volumetric fl ask and adjusted with acetonitrile, ethanol, and methanol (Merck, Darmstadt, Germany) to a fi nal volume of 50 ml individually. The solution (5 ml) was
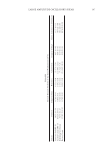
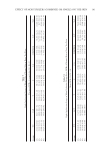
A VALIDATED OMC ANALYSIS: SPECTROPHOTOMETRIC METHOD 169 transferred into another volumetric fl ask (50 ml), and diluted into 5 μg/ml with the dif- ferent solvents. An absorbance pattern of each solution was recorded during 200–400 nm. The best solvent affording maximized absorption without the presence of interference effects was obtained. The stock OMC solution (0.8 mg/ml) was prepared into serial dilu- tions (4, 8, and 12 μg/ml), which were further validated on linearity, range, accuracy, and precision of the analysis. Linearity, range, accuracy, and precision. The above mentioned validated parameters were assessed at fi ve different concentrations (4, 6, 8, 10, and 12 μg/ml), and the absorbance was recorded at 310 nm. The linearity was determined with a determination correlation (r) of more than 0.995, in addition to a recovery (%) calculation as well as a relative stan- dard deviation (%RSD). All of the experiments were performed six times. VALIDATION OF OMC SUNSCREEN ANALYSIS METHOD Specifi city. The cosmetic bases (8 g) were weighted into volumetric fl asks (100 ml), sepa- rately, prior to the addition of methanol to achieve the fi nal volume. The solution was fi ltrated through Whatman no. 5 fi lter paper. The fi ltered solution (5 ml) was pipetted into 50 ml volumetric fl ask and diluted into 5 μg/ml. The absorbance was determined as described in the specifi city of OMC analysis. Accuracy and precision. The cosmetic bases (8 g of 1% OMC, 2 g of 4% OMC, and 1.07 g of 7.5% OMC) were weighted into six volumetric fl asks (100 ml). The stock OMC solution (0.8 mg/ml, 10 ml) in each fl ask was adjusted to 100 ml with methanol and mixed thor- oughly. The solution was fi ltered and diluted into 5 μg/ml. The absorbance determination was done at 310 nm as mentioned earlier. The procedure was conducted in six replicates. Repeatability of the method. OMC sunscreens in O/W and W/O emulsions, and mineral oil at different concentrations were weighted differently into the 100 ml volumetric fl ask Table I Ingredients of O/W and W/O Emulsions Emulsion Ingredient % (w/w) O/W Isopropyl myristate 12.0 Isopropanol 3.0 OMC 1.0 or 4.0 or 7.5 Stearic acid 1.0 Cetyl alcohol 0.5 Triethanolamine 1.0 Carbopol 934 0.2 Deionized water qts. to 100 W/O Petrolatum 17.0 Emulsifying wax 4.0 Calcium stearate 2.0 OMC 1.0 or 4.0 or 7.5 Caprylic/capric triglyceride 10.0 Jojoba 10.0 Microcrystalline wax 6.0 Deionized water qts. to 100
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)