18 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table I. Absorbances at 625 nm of 4 batches of F D & C Blue No. 1 (Concentration 5.0 x 10-4% w/v) Batch Absorbance Relative purity 1 O.726 100.0 2 0.699 96.4 3 0.688 94.8 4 0.693 95.5 as to compensate for dye purity in the production process. Should the batch need subsequent correction, the following actual examples will illustrate the process. For practical purposes, the absorption coefficient is expressed in terms of the so-called E one-one (E•0, this being the (imaginary) absorbance of a 1 •o solution of the absorbing species in a cell of 1 cm thickness. Example 1--Spectral match, single colour Product Composition Wavelength of maximum absorption of dye Absorbance of standard at 625 nm Absorbance of sample at 625 nm E• of dye at 625 nm Visual assessment Problem Cologne Perfume base + blue dye 625 nm 0.224 units 0.186 units 1172.1 units Sample weak in blue and in yellow How much dye must be added to a 450-litre batch to obtain a match? Required increase in absorbance = 0.224--0.186 = 0.038 units. Since a 1 •o concentration increase would give a change of 1172.1 units, the required increase is: 0.038 1 x 1172.• - 3.24 x 10-5•o. To increase the concentration of dye by this amount in 450 litres requires: 3.24 x 10 -5 x 4.5 x 10 a = 0.1458 g. Result: The addition of 0.146 g of dye gave an absorbance of 0.226 units
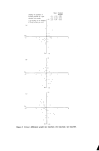
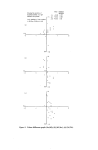
INSTRUMENTAL COLOUR ASSESSMENT 19 .. Yellow (a) .......... Base ........... Yel low .......... Red ............. Green i, I ß \ Green "--• 4 ,5 6 Wavelength nm x 10 -2 Figure ](a). Absorption spectra of aftcrsha¾c components, (b) Absorption spectrum of an aftershave. (b) 7.5
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)