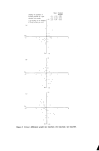
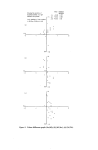
20 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS at 625 nm. Visual assessment was a good match in blue, but sample still slightly weak in yellow. We concluded that the physical standard had 'yellowed' with age. If we accept that the perfume base plus its various additives will in- evitably get darker with age, there seems nothing for it but to abandon 'real' colour standards in favour of stable metameric substitutes, or to define the colour standard in terms of instrumental readings. We have adopted the latter with some success. The absorption spectra of multi-dye products are obtained simply by adding together the spectra of the individual dyes at their relevant concen- trations. In other words, the absorbance at any wavelength of the final product will be Ax• = Kx•CL + KI•iC1L + K11MC11L + ............. where K, K 1, K 11 and C, C 1, C 11 refer to individual dyes. At another wave- length •..,. Axe. = Kx•CL -]-Kl•CiL-]-K11•C11L -['- .............. Fig. l(a) and l(b) illustrate this additive phenomenon. The absorption spectrum of an aftershave product is obtained by the algebraic addition of the spectra of three dyes in suitable concentrations plus a contribution from the perfume base. Three measurements are taken at points on the spectrum where the absorbance is predominantly due to each of the controllable variables--one each at the maxima of the red and green components (532 and 642.5 nm, respectively) together with a point on the yellow curve where the absorption by the base does not interfere (470 nm). Example 2--Spectral match, three colours Product Composition Wavelengths of maximum absorption of dyes: red green yellow Aftershave lotion Perfume base plus three dyes red, green, yellow 532 nm 642.5 nm 425 nm (measured at 470 nm)
INSTRUMENTAL COLOUR ASSESSMENT 21 Absorbance of standard at 532 nm 642.5 nm 470 nm E• of dyes Red Green Yellow Problem At 470 nm, 0.152 units 0.140 units 0.314 units 470 nm 532 nm 642.5 nm 183.9 507.8 22.9 49.7 176.4 445.5 14.7 What concentration of dyes having the above E•x values would be needed to match the standard? absorbance 0.314 = (183.9 Cr) + (22.9 Cg) + (445.5 Cy) At 532 nm absorbance 0.152=(507.8 Cr) +(49.7 Cg)+(14.7 Cy) At 642 nm absorbance 0.140 = (176.4 Cg). Where Cr, Cg and Cy are the concentrations, expressed in percent, of the red, green and yellow dyes respectively. From these simultaneous equations. Cg -- 7.936 x 10-4•o wv -4 Cr = 2.048 x 104•o wv -• Cy --- 5.794 x 104•o wv -• Result: A trial 10-litre batch of aftershave was made incorporating the above concentration of dyes. Absorbance at 470 nm = 0.305 units 532 nm = 0.157 units 642.5 nm = 0.141 units Visual assessment: a very good match. Three-component colour matching problems such as this pose consider- able difficulties for the unaided human assessor. The introduction of this simple instrumental procedure dramatically reduced the correction time for this product at our factory. The assessment of the purity of soluble dyes and their concentrations in clear liquid products by spectrophotometer has been practised for a number of years in the authors' laboratory with considerable success. In practice, each batch of product is arranged to be slightly too strong in colour so that a single correction can be calculated involving the addition
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)