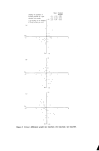
22 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS of solvent this takes care of any slight manufacturing variations which, it seems, are impossible to eliminate completely. REFLECTANCE SPECTROPHOTOMETRY Having achieved success in dealing with clear, coloured, liquid products, we were anxious to test the spectrophotometer's performance in the assess- ment of coloured solids and opaque liquids. For this we required a reflect- ance attachment which modifies the light-path through the instrument so as to allow the examination of light reflected from the surface of the sample rather than that passing through it. Several designs of reflectance device were tried and that finally chosen was of the integrating sphere variety, since this showed less variation of output with sample orientation than did any of the others, especially for pearly products. Fig. 2 illustrates the form in which data is obtained from a reflectance spectrophotometer. The reflectance curves A, B, C and D were obtained from lipstick components and represent a measure of the percentage of incident light which was reflected at each wavelength for each component. It can easily be seen that the pigments A and B reflect mainly in the red region (A being more orange than B). D reflects light over most of the -- I diwslon = I0 % R ..... Orange-Red ..... Red .......... Brown ....... Base ß ß ß / // ß ß Blue Green Yel low Red :5.5 4- 5 6 7.5 Wavelength nm x 10 -2 Figure 2. Reflectance spectra of lipstick components.
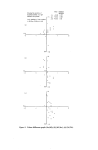
INSTRUMENTAL COLOUR ASSESSMENT 23 visible spectrum and is yellow in appearance whilst C is not a good reflector, but absorbs less in the red and orange regions and is therefore dark brown in colour. Although there is a simple quantitative relationship between the con- centration of a colour and its percentage reflectance at a given wavelength (as in absorption spectrophotometry) we soon found that the combinations of similar dyes and the relatively large number of components contributing to the colour of the final solid product made any simple quantitative treat- ment of colour correction both time consuming, and imprecise when the instrument was used in the reflectance mode. Moreover, the amount and quality of light reflected from a surface depends not only on the colour of the individual components but also upon their distribution and particle size as well as the depth of gloss and refractive index of the surface itself. Nevertheless, we were interested to discover to what extent discernible differences in the observed colour of two similar products were mirrored by their reflectance spectra--in the hope that such data could be used as invariable standards. Fig. 3 illustrates the spectra of two samples of the same shade of lipstick. The samples were prepared by melting the lipstick into a 27 mm godet and flaming the surface to a smooth, even finish. Curve A was obtained from •i,• 'f I division = I0% R -- ] - .• BA Standardø90rataled•i•½½StandardSample / • •L.•_• ....... /•,i, C /•/ •(%. /•',t u Sample rotated 90 ø ½ .½' '%t"-•.- A B..½½r½'/,,' • D C 55 4 5 6 Wavelength nm x10 -• Figure 3. Reflectance spectra of a lipstick. 7.5
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)