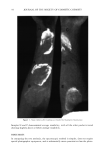
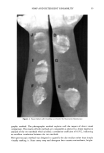
•J. Soc. Cosmet. Chem., 37, 49-57 (March/April 1986) A three-day mold assay for cosmetics and toiletries CATHERINE A. MEAD and JOHN J. O'NEILL, Avon Products Inc,, Division Street, Suf•rn, NY 10901, Received January 6, 1986. Synopsis Cosmetic product mold recovery assays generally call for five or more days of incubation of a nutrified agar plate in which the product has been dispersed. This paper reports the development and validation of an assay procedure which requires only three days of incubation. It is a refinement of the five-day test in two respects. First, and most importantly, the temperature is carefully controlled in an incubator at 27 to 28øC, replacing "room temperature." Second, a more nutritious agar, buffered near neutrality, replaces the low pH Mycophil agar (BBL) used in our conventional five-day test. The method was validated by simulating contamination of a wide variety of cosmetic products with environmental and ATCC fungal strains. BACKGROUND INCUBATION TEMPERATURE The rate of mold growth is strongly temperature-dependent (1). Over the range of 0 to 40øC, it increases with increasing temperature to a maximum and then decreases abruptly to a negligible value just a few degrees above the maximum (2-5). The tem- perature of maximum growth rate differs from one species to another, over a range of 20 to 35øC. Incubation temperature is critical for any mold species when growth rate is to be opti- mized. The optimum temperature can be readily determined for any given mold species, but for the wide collection of genera likely to be encountered as contaminants of cosmetics, it is obvious that a compromise must be made. PRESERVATIVE CARRYOVER AND AGAR pH When a product is assayed for contamination it is quantitatively diluted tenfold into an aqueous broth and then diluted about tenfold further when the broth is dispersed in the agar growth medium. In the agar all the components of the product have been diluted about one hundred fold. Cosmetic ingredients, including preservatives such as benzoic acid, may interfere with mold spore germination or growth even after dilution. Benzoic acid is the major preservative in many shampoos and bubble baths, usually at a level of 0.2%. Dilution of the product with broth and agar reduces its final concentration to about 0.002%. Previous work in this laboratory with benzoic and sorbic acids had shown that they are active at levels of a few thousandths of one percent if the pH is appreciably below 6, but not active near neutrality. Thus, benzoic acid carryover might 49
50 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS inhibit growth in low pH Mycophil agar (BBL) (pH ca. 4.7), but not in an agar which is buffered near neutrality. Less interference was expected from other preservatives. Methyl paraben at 0.8% in product (the maximum level recommended in U.S.) is diluted to less than 0.01%. This is about one twentieth of saturation in water, a level which reduces the growth rate of Escherichia co/i by about 10% (as found in previous studies in this laboratory) and might have a comparable inhibitory effect on mold growth. This effect would be diminished by the solvent action of Tween 80 (ICI Americas, Inc.) in the agar. Other preservatives and co-preservatives such as alcohols and glycols were not expected to interfere more than methyl paraben. MATERIALS AND METHODS PRELIMINARY RESEARCH In general, mold spore assay methods require five or more days incubation (6-9). In our preliminary research with Aspergillus niger ATCC # 16404 (one of the strains used in challenging new products during the developmental cycle), it was found that incu- bation for three days at room temperature yielded complete counts of mycelial colonies several millimeters in diameter after five days the colonies were larger but no more numerous. In most cases, there were complete counts after only two days, and occasion- ally the number of colonies recovered after incubation for only one day was the same as after five. We began to suspect while studying other variables that high growth rate coincided with unusually high room temperature and that this uncontrolled variable (uncontrolled in spite of typical industrial summertime air conditioning) was distorting the response to well controlled changes in the nutrients added to the agar. A few experiments with crude temperature control at about 32øC in an ordinary convection oven supported this idea at this higher temperature complete counts were observed after 24 hours. Good temperature control near room temperature calls for both heating and refrigera- tion. Most incubators offer only heating units and these cannot achieve accurate temper- ature control below 30øC. Good control is provided in Biological Oxygen Demand (BOD) cabinets (Precision Model 815, Forma Scientific Model 3770, or equivalent) designed to maintain temperatures at and below ambient. Using this equipment, we measured the growth of A. niger ATCC #16404 and three other strains at 20, 25, 30 and 35øC in four agars, each containing 2% Tween 80: Mycophil BBL# 11445 (referred to as MYN), Mycophil with low pH BBL#11450 (MYA--the agar used at Avon Products, Inc. in the standard five-day mold assay), HCA, and HC. The last two media are yeast-salts agars buffered at pH 4.6 and 6.7 respectively (Table I). AOAC Letheen broth BBL# 10914 was used as the diluent in all experiments. The neutral HC agar contains 100 ppm chloramphenicol to prevent bacterial contami- nation. At this level, challenges with strains of Pseudomonas aeruginosa and Serratia mar- cescens (which grow on MYA) are completely eliminated. At 200 ppm, twice the level in HC agar, chloramphenicol has no effect on the growth of any of the eleven mold strains listed in Table II.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)