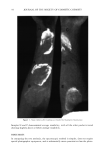
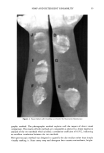
j. Soc. Cosmet. Chem., 37, 99-104 (March/April 1986) Validation of the agar patch test using soap bars which deposit different amounts of triclocarban FRANK YACKOVICH, NEIL K. POULSEN, and JOHN E. HEINZE, The Dial Corporation, Dial Technical Center, 15101 N. Scottsdale Road, Scottsdale, AZ 85254. Received November 18, 1985. Synopsis The agar patch test is a new method for evaluating the bacteriostatic activity deposited on panelists' forearms by washing with antibacterial soap bars. The test involves placing bacteria-streaked agar plates against washed forearms followed by measurement of bacterial survival on the plates after incubation. In an attempt to validate the test method, soap bars depositing different amounts of triclocarban were compared. Previous unpublished results showed that Soap B (0.8% triclocarban, new formulation and processing method), although it contained less triclocarban than found in Soap A (1.5% triclocarban, old formulation and processing method), deposited more triclocarban on skin. Consistent with the deposition results, the agar patch test showed significantly better residual bacteriostatic activity on forearms after one washing with Soap B than with either Soap A (1.5% triclocarban) or Soap C (placebo--no triclocarban). These differences were significant at the .01 probability level. This result would be missed in an in vitro test such as the Minimum Inhibitory Concentration test. The agar patch test also showed that there was no apparent trend for the number of washings to affect the ratio of activity of Soap B to Soap A. Apparently the new formulation and processing method used to make Soap B increased the amount of triclocarban deposited but not the rate (amount per washing) at which triclocarban was deposited. INTRODUCTION An in vivo agar patch method was developed (1) to evaluate the substantivity and bac- teriostatic activity of soap bars on human skin. This improved method was a modifica- tion of the method of Eigen et al. (2). The modified test involved placing bacteria- streaked agar plates against washed forearms followed by measurement of bacteria sur- vival on the plates after incubation. This modification eliminated the need for the bacteriostat to diffuse into the medium. For a bacteriostat such as triclocarban (3,4,4'- trichlorocarbanilide), which has very low water solubility, diffusion into the media was too slow to prevent overnight bacterial growth. In the development of the in vivo agar patch test, a deodorant soap containing 1.5% triclocarban was compared to a placebo soap (no active agent). It was shown that the residual bacteriostatic activity of the deodorant soap-washed forearms compared to the placebo soap-washed forearms reached a maximum after 7 washes and remained at that level at least through 13 washes (1). The question remained, however, as to whether the agar patch method could distin- guish differences in residual bacteriostatic activity between soap bars which deposited different amounts of bacteriostat. Two soap bars with the same base, different amounts 99
100 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS of triclocarban, and different formulation and processing methods were used to answer this question. Theiler and Sands (3) previously showed that Soap B (0.8% triclocarban, new formulation and processing method) deposited 2 to 6 times more triclocarban on depilitated rat skin than Soap A (1.5% triclocarban, old formulation and processing method). Apparently the new formulation and processing method used for Soap B al- lowed more triclocarban to deposit on skin even at lower concentrations of triclocarban in the soap bar. Thus the purpose of the experiments reported here was to validate the agar patch test with soap bars known to deposit different amounts of triclocarban on skin and to con- firm that Soap B deposited more triclocarban on skin than did Soap A. EXPERIMENTAL SOAP BARS Soap A (old formulation and processing method) contained 1.5 % triclocarban in a base of sodium tallowate, sodium cocoate, water, glycerine, fragrance, sodium chloride, preservatives, and color. Soap B (new formulation and processing method) contained 0.8% triclocarban in a base of sodium tallowate, sodium cocoate, water, PEG-6 methyl ether, fragrance, sodium chloride, glycerine, preservatives, and color. Soap C (a com- mercial product) was analyzed and found to contain sodium tallowate, sodium cocoate, potassium tallowate, potassium cocoate, sodium chloride, alcohol insolubles (inorganic material), glycerine, magnesium silicate, chelating agent, and fragrance, but did not contain a bacteriostat. AGAK PATCH TEST METHOD The agar patch test method has been described in detail previously (1). A brief sum- mary of the method used in this study follows: A third consecutive overnight culture of Staphylococcus epidermidis ATCC 155 was diluted 6/10,000 and a 4-ram loopful (400 colony-forming units, CFUs) was spread evenly over the convex surface of solidified and 24-hr-dried mannitol salt agar (Difco) in 35 x 10-ram petri dishes (without lids). After completing a 1-week period without using deodorant soaps, panelists washed their forearms with test bars under supervision with at least a 2-hr drying out time between washes. During the drying out time the panelist resumed normal activities, but avoided chemical or water contact with the forearms. Thirty minutes after the last wash, the inoculated surfaces of three streaked and dried agar plates were attached firmly to each forearm using tape. The plates were removed after 30 minutes or two hours contact time and were incubated for 24 hrs at 35øC to allow growth of surviving test bacteria. Colonies were counted using a 10 X dissecting microscope. CALCULATIONS For the comparison of the three soap bars, each of the following applications were assigned to the same number of panelists:
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)