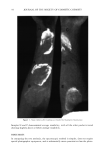
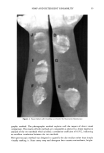
THREE-DAY MOLD ASSAY 51 Table I Agar and Nutrient-Buffer Formulations HCA Agar (yeast-salts, low pH) Glucose (Dextrose) 20 g Agar 15 g Yeast extract (BBL ! 1922) 5 g Polypeptone (BBL 11910) 5 g Nutrient-buffer salts solution, pH 4.6 (see below) 25 ml Distilled water 975 ml Tween 80 (ICI Americas) 20 g HC Agar (yeast-salts, neutral pH)* Glucose (Dextrose) 20 g Agar 15 g Yeast extract (BBL ! 1922) 5 g Polypeptone (BBL 11910) 5 g Nutrient-buffer salts solution, pH 6.7 (see below) 25 ml NaOH solution, 4N 6 ml Chloramphenicol-1000 ppm solution in water 100 ml Distilled water 869 ml Tween 80 (ICI Americas) 20 g Nutrient-Buffer Salts Solution, pH 4.6 DL-Malic acid 1.0 M K2HPO 4 0.4 M NH4C1 1.0 M MgSO4 0.01 M NaOH 1.0 M Nutrient-Buffer Salts Solution, pH 6.7 Na2HPO4 1.0 M KH2PO 4 1.0 M NH4C1 1.0 M MgSO4 0.01 M * A complete, dehydrated medium (excluding Tween 80) now available from BBL (Catalog #97110). CONTROL OF MOISTURE LOSS In the course of this work, it was noted that agar incubated in the B. O.D. cabinets was cracking and shrinking apart much more than at "room temperature." Measurements Table II Fungal Strains Organism Source 1. Geotrichum fici 2. Aspergillus niger 3. Cladosporium resinae 4. Fusarium accuminatum 5. Aspergillus species 6. Aspergillus oryzae 7. Penicillium notatum 8. Aspergillus spedes 9. Aspergillus niger 10. Aspergillus species 11. Aspergillus species ATCC 34015 (Type Culture) ATCC 16404 (U.S. Pharmacopeia) ATCC 11273 (from ointment) ATCC 32949 (from jet fuel) 6177B (in-house isolate) ATCC 10196 (fungus resistance testing of paint) ATCC 9478 (culture contaminant) 10,337 (in-house isolate) BL-89 (Institute of Paper Chemistry) 6929 (in-house isolate) 6871 (in-house isolate)
52 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS on individual dishes showed weight losses as high as 65% in three days. Presumably this would inhibit growth because it would triple the concentrations of dissolved solids in the remaining water, including preservatives carried over with the product. After trials of several variants, we adopted the procedure of cooling the dishes on the bench top for about an hour after pouring and then returning them to the plastic bags (in which they are received from the manufacturer), punctured every few inches (with a sharp pencil) for venting. The bags were sealed with tape and placed in the incubator inverted, to reduce lid condensate. Under these conditions weight losses ranged from 1 to 4% in three days, and 1 to 7% in five days lid condensation was reduced to a tolerable level. A COMPARISON OF FUNGAL GROWTH IN SEVERAL MEDIA Using an incubation temperature of 27 to 28øC with plates stacked inverted in punc- tured bags, the eleven mold strains shown in Table II were grown in six agars, all with 2% Tween 80: MYA, MYN, Potato Dextrose BBL# 11550, Potato Dextrose with tar- taric acid added, and the yeast-salts agars, HCA and HC. Colonies were counted and their diameters measured on Days 1, 2, 3 and 4. SIMULATED ASSAY OF MODEL PRODUCTS Model products used in this study were a shampoo base [10% sodium lauryl sulfate (SLS), 5% lauroyldiethanolamide (LDA) and phosphoric acid to adjust pHI, talc without additives, and a simple emulsion of 20% peanut oil in water with 3.5% non- ionic emulsifiers, Myrj 52 and Brij 72 (ICI Americas, Inc.). These three "products" could have very different effects on the clarity of the agar and thus the readability of the plate count results, i.e., no effect with the shampoo, maximum turbidity with the talc, and an intermediate effect with the emulsion. One gram of "product" was dispersed in 9 ml broth containing about 100 or 300 spores, thus simulating assay of products contaminated with 100 or 300 spores per gram. The spores were harvested from cultures grown for 7 to 10 days into 0.1% saline with 0.1% Tween 80 (diluent was added to the plate and spores loosened with a swab) and diluted with broth to about 100 spores per mi. One milliliter of the broth dispersion was mixed with cooled molten agar (MYA or HC) in a petri dish (delivering 0.1 g of product and 10 or 30 spores). The dishes were handled and incubated as described above. In all experiments, a dispersion of spores in broth was prepared as a control for each mold strain. Plates were prepared in duplicate from each dispersion. The plates were examined at three and five days. VALIDATION IN PRODUCTS A simulated assay of 85 cosmetic products was carried out as described above. This validation procedure was generously conservative in comparing the proposed three-day method with the standard five-day method because it gave the latter the benefits of well controlled optimum incubation temperature in BOD cabinets (rather than "room tem- perature" previously used in this laboratory) and protection against excessive moisture loss.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)