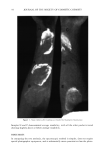
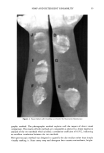
90 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS evaluating wash tests of the face and antecubital areas produced several changes in results compared to the earlier soap chamber test (6). One possible explanation of the difference in results between these methods may be the nature of the test procedures used. The soap chamber test is basically a repeated oc- cluded patch test which does not allow for rinsing of the soap from the skin, an integral part of the use of any cleansing product such as soap. Wash testing, which includes rinsing, more closely approximates actual consumer use. This raises the question of the importance of rinsability as an attribute affecting the mildness of soap products. As- suming that the longer a soap remains on the skin, the greater the potential for irrita- tion, a number of cleansing bar products were investigated solely for their rinsabilities. MATERIALS Eighteen common soaps, combars, and syndet bars were obtained from local retail stores (List 1). The bars were cut into fine shavings and dissolved in tap water to a final concentration of 10% (w/v). Tap water (260 ppm hardness) was selected for these ex- periments since it most closely simulates actual consumer water hardness levels. Fluo- rescein dye (Sigma, St. Louis, MO) was added to obtain a dye concentration of 0.005% (w/v) and the mixture thoroughly stirred. Similarly, a 10% reference solution of each bar was prepared in the same manner without fluorescein dye. INITIAL EXPERIMENTS Fluorescein dye was initially evaluated by placing a 1.0% dye solution on the forearms of volunteers and allowing this to stand for 15 minutes. The arms were then rinsed and examined under ultraviolet light to determine if the dye was in any way substantive or absorbed into the skin. It was found that a 1.0% fluorescein dye solution left no detect- able residue on the skin after 15 minutes. Further studies indicated that only after 45-60 minutes were detectable amounts of fluorescein dye adsorbed into the skin. To demonstrate the proportionality of the fluorescein dye to the amount of soap residue, 5.0 ml of a 10% soap/fluorescein dye solution was applied to each of 10 tared etched glass plates (8 cm x 8 cm). The solutions were then worked into a lather and rinsed gently with 100 ml of tap water. The plates were then allowed to dry at 50øC to a constant weight, and the amount of soap residue was then calculated. Each plate was then extracted with 50 ml of methanol:water (80:20) and the amount of fluorescein dye quantified spectrophotometrically at 280 nm. The ratio of the initial soap-to-dye con- centration was then compared to the ratio of the soap residue (weight) to the residue dye concentration and were found to be equivalent. (Table I). This experiment was repeated with three other soaps of varying rinsabilities, with the same results. (Table I). From this, we determined that the amount of fluorescein dye deposited with the soap residue is proportional to the amount of soap left on the skin. PHOTOGRAPHIC METHOD A small cloth was saturated with a fluorescein-containing soap solution. The solution was applied to a randomly chosen site (approximately 5 cm in diameter) on the ventral aspect of the forearm of a volunteer, using up to three different soap solutions per
SOAP AND DETERGENT RINSABILITY 91 List 1 Product Code by Ini•redient Predominance A: Non-detergent, mineral oil, lanolin, PEG-4 dilaurate B: Collodial oatmeal, lanolin derivative, mild surfactant C: Sodium tallowate, sodium cocoate, stearic acid D: Sodium tallowate, sodium cocoate, water, glycerin (active ingredient: triclocarban) E: Sodium cocoyl isethionate, stearic acid, sodium tallowate, water, sodium isethionate F: Sodium tallowate, sodium cocoate, stearic acid G: Triethanolamine, sodium tallowate, stearic acid, glycerin H: Sodium and potassium fatty acids, glycerin I: Sodium tallowate, sodium cocoate, water coconut acid (active ingredient: triclocarban) J: Sodium tallowate, potassium lauryl sulfate, magnesium tallowate (active ingredient: triclocarban) K: Sodium cocoyl isethionate, stearic acid, dextrin, water, sodium isostearoyl-2- lactylate. L: Sodium cocoate, sodium tallowate, coconut acid, water M: Sodium cocyl isethionate, stearic acid, sodium tallowate, water, sodium isethionate N: Sodium tallowate, sodium cocoate, coconut acid, water O: Sodium cocoyl isethionate, stearic acid, dextrin, water, sodium isostearoyl-2- lactylate P: Sodium tallowate, sodium cocoate, water, coconut acid Q: Stearic acid, rosin, coconut oil, water, glycerine R: Sodium tallowate, sodium cocoate, water, glycerin, cocoa butter Z: Sodium tallowate, potassium lauryl sulfate, magnesium tallowate, sodium lauryl glyceryl sulfate, water (active ingredient: triclocarban) forearm. The soap solution was worked into a lather for approximately 15 seconds with a circular motion using the applicator cloth. After the application was completed, each site was rinsed with 250 ml of room temperature tap water, and either air-dried or gently blotted dry. The soap/fluorescein residues were photographed using Ektachrome 400 film with an aperature setting of f8. The arms were illuminated using a Novatron strobe equipped with a 18A ultraviolet filter. A total of ten soaps were analyzed using the photographic method. The relative rins- ability was then determined by a comparison of the photographic intensities of the fluorescein dye residue as ranked from 1 to 10, 10 being the greatest residue, as rated by three independent judges. These ratings were averaged to form an overall ranking.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)