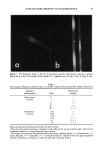
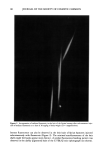
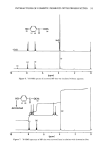
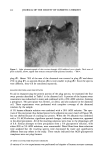
122 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS larly interesting. This band also appeared in the ACH-albumin (Figure 4C) and ACH- sweat protein (Figure 4D) precipitates, which further indicates a strong interaction between ACH and stratum corneum (Figure 7). Although somewhat controversial (18), this band is usually assigned to a metal-coordinated NH 2 deformation mode (11, 12) in metal-amino acid complexes with metal-nitrogen covalent bonds at frequencies between 765 and 790 cm-•. Further, the negative bands near 3300 cm- • in Figure 7 can be assigned to N-H stretch vibrations, demonstrating purturbation around that functional group. These two assignments, plus the shift in the amide B band (also N-H related) described earlier for the plug #2 spectrum (Figure 5A), are good evidence for alu- minum-nitrogen coordination. Whether this coordination occurs directly or via inter- mediate hydroxyl ligands is not distinguished. CONCLUSION Vibrational spectroscopy has been useful in characterizing areas of poral occlusion (plugs) from ACH-treated forearms of two different subjects. Even though the two plug spectra were different, good evidence was found in each for the formation of a complex between aluminum and stratum corneum. One plug spectrum was consistent with ACH-stratum corneum protein interactions plus some free ACH precipitate. The spec- trum from the second plug was a close match with another obtained in vitro by soaking prehydrated axillary stratum corneum in ACH. The protein portion of both plugs was spectroscopically similar to ACH-treated axillary stratum corneum even though the plugs were dissected from forearm biopsies. Perhaps the composition of the keratinized portion within the distal part of any duct is always axilla-like. This ACH-keratin com- plex was distinguished from sweat proteins or ACH-sweat protein precipitates and in- dicates that sweat proteins are not a component of the emphractic material. Binding to keratin was strong and irreversible. It is well established that the upper portion of the intra-epidermal sweat duct is keratin-lined (3,6,26). Epidermal cells migrate laterally toward the ductal lumen and ring the lumen with keratinocytes which actively keratinize and desquamate into the lumen. Thus, within the lumen a supply of keratinized cells is readily available for ACH binding. The hydration state of these cells should be high, a condition which favors aluminum binding. Our spectral evidence, therefore, agrees in effect with the keratin supercontraction hypothesis proposed by Shelley and Hurley (3). A synergistic effect, partially due to the formation of a metal- hydroxide gel emphraxis in the sweat duct [proposed by Relier and Luedders (4)], cannot be totally ruled out. Evidence was found in plug # 1 for the presence of free ACH (band near 1080 cm-1). The spectrum from plug #2, however, showed alu- minum only in the bound state (to keratin), as evidenced by a strong band at 1049 --1 cm It is logical to expect that chemical binding to keratinized ductal walls is required to stabilize a plug and offer resistance to displacement by hydraulic pressure within the duct. This is especially true for ACH, the site of action of which is rather superficially located in the distal portion of the duct within the horny layer. Frictional forces will act to maintain a free forming precipitate (or sweat-protein complex), and such blockage might be expected to float free or be flushed away during firing of a thermally stressed duct.
FTIR OF SWEAT GLANDS & ALUMINUM SALTS 123 In their review, Shelley and Hurley (3) point out that virtually any metallic salt can be expected to show a topical antiperspirant effect. Further, the binding of virtually all metal salts to many proteins and polypeptides is well established (25). Is this the common thread between antiperspirant efficacy and metal salts? Relier and Luedders (4) showed an inverse relationship between antiperspirant efficacy and the affinity of the ligand to form an aluminum complex in a series of aluminum salts. Aluminum salts of strong complexing agents (i.e. citrate) were found to be completely ineffective as anti- perspirants. In our laboratory, precombination of ACH with other species led to re- duced antiperspirant efficacy when compared against ACH alone (28). An explanation for both of these effects lies in the reduced availability of the aluminum to bind to stratum corneum proteins because of precomplexation with other ligands or additives. All of these findings implicate the coordination of ACH with stratum corneum as a vital part of the mechanism of antiperspirant action. REFERENCES (1) W. B. Shelley and P. N. Horvath, Experimental millaria in man. II. Production of sweat retention, anhidrosis and miliaria crystallina by various kinds of injury, J. Invest. Dermatol., 14, 9-20 (1950). (2) W. B. Shelley and P. N. Horvath, Experimental miliaria in man. III. Production of miliaria rubra (prickly heat), J. Invest. Dermatol., 14, 193-204 (1950). (3) W. B. Shelley and H. J. Hurley, Studies on topical antiperspirant control of axillary hyperhidrosis, Acta. Dermatovener (Stockholm), 55, 241-260 (1975). (4) H. H. Relier and W. L. Luedders, "Pharmacologic and Toxicologic Effects of Topically Applied Agents on the Eccrine Sweat Glands," in Advances in Modern Toxicology, Dermatotoxicology and Pharma- cology, F. N. Marzulli and H. I. Maibach, Eds. (Hemisphere Publishing Company, Washington and London, 1977), Vol. 4, pp. 1-5. (5) E. Holzle and A.M. Kligman, Mechanism of antiperspirant action of aluminum salts,J. Soc. Cosmet. Chem., 30, 279-295 (1979). (6) R. P. Quatrale, D. W. Coble, K. L. Stoner, and C. B. Felger, The mechanism of antiperspirant action by aluminum salts. II. Histological observations of human eccrine sweat glands inhibited by aluminum chlorohydrate. J. Soc. Cosmet. Chem., 32, 107-136 (1981). (7) R. P. Quatrale, A. H. Waldman, J. G. Rogers, and C. B. Felger, The mechanism of antiperspirant action by aluminum salts. I. The effect of cellophane tape stripping on aluminum salt-inhibited eccrine sweat glands, J. Soc. Cosmet. Chem., 32, 67-73 (1981). (8) G. J. Putterman, J. Strassburger, and J. J. Fitzgerald, In vitro sorption of aluminum complex to guinea pig stratum corneum,J. Invest. Dermatol., 77, 319-324 (1981). (9) C. E. Weir, E. R. Lippincott, A. Van Valkenburg, and E. N. Bunting, Infrared studies in the 1 I.tm to 15 I.tm region to 30,000 atmospheres, J. Res. Nat. Bur. Standards, A63, 55 (1959). (10) D. A. Reisgraf and M: L. May, Infrared spectra of aluminum hydroxide chlorides, Appl. Spectrosc., 32, 362-366 (1978). (11) D. Segnini, C. Curran, and J. V. Quagliano, Infrared absorption studies of inorganic coordinated complexes--XXIII. Studies of some metal alanine complexes, Spectrochim. Acta., 16, 540-546 (1960). (12) A. J. Saraceno, I. Nakagawa, S. Mizushima, C. Curran, and J. v. Quagliano, Infrared absorption spectra of inorganic coordination complexes. XVI. Infrared studies of glycino-metal complexes, J. Am. Chem. Soc., 80, 5018-5021 (1958). (13) K. Nakamoto, Y. Morimoto, and A. E. Martell, Infrared spectra of aqueous solutions. I. Metal chelate compounds of amino acids, J. Am. Chem. Soc., 83, 4528-4532 (1961). (14) I. Lyon and I. M. Klotz, The interaction of epidermal protein with aluminum salts, J. Am. Phar- macol. Assoc., 47, 509-512 (1958). (15) F. K. Habib, J. H. Keighley, P. Rhodes, and C. S. Whewell, The interaction ofmetal-amines with keratin--I. Infrared studies of the binding of Zn(II) to keratin, Spectrochim. Acta., 31A, 1-10 (1975). (16) J. Strassburger and I. T. Smith, Spectral subtraction using Fourier transform infrared spectroscopy-- fact or artifact, Appl. Spectrosc., 33, 283-286 (1979).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)