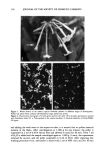
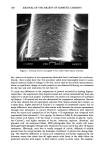
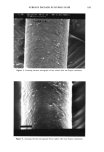
j. Soc. Cosmet. Chem., 44, 139-151 (May/June 1993) In vitro sun protection factor evaluation of sunscreen products KATHERINE A. KELLEY, PAUL A. LASKAR, GARY D. EWING, SYDNEY H. DROMGOOLE, J. LEON LICHTIN, and ADEL A. SAKR, Allergan Herbert, 2525 Dupont Drive, Irvine, CA 92715 (K.A.K., P.A.L., G.D.E.), College of Pharmacy, University of Cincinnati, 3223 Eden Avenue, Cincinnati, OH 45267-0004 (J.L.L., A.A.S. ), and Oclassen Pharmaceuticals, Inc., 1 O0 Pelican Way, San Rafael, CA 94901 (S.H.D.). Received June 9, 1992. Synopsis An in vitro method to evaluate sunscreen products for static sun protection factor (SPF) was investigated. Surgical adhesive tapes were used as substrates in a procedure modified from that originally described by Diffey and Robson, 1989. The modified technique utilized a 150-watt xenon arc solar simulator and an ultraviolet B radiometer. The in vitro SPF was calculated as the ratio of light transmission through untreated tape to that of sunscreen-treated tape and compared to SPFs determined in vivo. Correlation between in vivo and in vitro SPF data varied tremendously. The sources of error were attributed to the structure of the tape, absorption of the sunscreen active ingredients into the tape, and the detection limit of the radiometer used. INTRODUCTION Predictive in vitro methods to evaluate SPFs of sunscreen formulations prior to clinical testing would be extremely useful tools for formulators. Ideally, such test procedures should be easily executed in a timely manner, utilizing readily available testing mate- rials, and should provide reproducible, accurate results that correlate with in vivo SPF data. A number of papers related to the development of in vitro techniques for assessing the photoprotection of sunscreen formulations have been published. Substrates for these methods included wool (1,2), pig skin (2), mouse epidermis (3-6), synthetic skin casts (7,8), and surgical tape (9). Ultraviolet light detection systems have included integrat- ing sphere spectrophotometers (3,4), scanning spectroradiometers (9), and radiometers with detector responses that are similar to the human erythemal action spectrum (5,6). The surgical adhesive tape method developed by Diffey and Robson appeared to provide a straightforward technique to estimate SPF in vitro, utilizing a medical adhesive tape and a scanning spectroradiometer (9). The authors commented that the methodology could be adapted to other experimental setups. The present research investigated the potential use of two medical adhesive tapes and an experimental setup similar to that 139
140 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS used by Cole and Van Fossen (5,6) to estimate in vitro the SPFs of several sunscreen formulations. MATERIALS AND METHODS TEST PRODUCTS The sunscreen products used consisted of commercial sunscreens, investigational prod- ucts, and the FDA homosalate standard sunscreen. Table I lists the active ingredients and formulation types of each of the sunscreens tested in this study. The estimated in vitro SPF of each sunscreen was determined in triplicate on each of the tape substrates. Table I Comparison of In Vivo and In Vitro SPFs Label SPF Sunscreen Active Formulation or in In vitro SPF** In vitro SPF** product ingredients type vivo SPF Transpore © tape Blenderm © tape A Titanium dioxide Creamy lotion 15 4.17 + 0.99 15.94 + 4.39 B Benzophenone-3 Lotion 18' 31.25 + 9.2 23.82 + 3.74 Avobenzone Octyl salicylate Octocrylene C Homosalate FDA lotion 4.2 3.3 + 0.92 4.5 + 1.15 D Octyl dimethyl PABA Lotion 15 (17') 27.42 + 5.31 40.37 + 3.73 Avobenzone E Octyl methoxycinnamate Alcohol gel 25 17.13 + 5.98 57.6 + 51.92 Octyl salicylate Homosalate Benzophenone-3 F Octyl methoxycinnamate Oil spray 5 9.22 + 3.55 22.13 + 6.27 Benzophenone-3 Menthyl anthranilate G Octyl methoxycinnamate Lotion 17.4' ND*** 8.61 + 2.4 Octyl salicylate Benzophenone-3 H Titanium dioxide Lotion 10 ND*** 6.81 -+ 1.8 J Titanium dioxide Lotion 8 ND*** 8.94 + 1.38 K Titanium dioxide Lotion 15 ND*** 349.9 + 316.0 L Titanium dioxide Cream 25 ND*** 235.56 + 40.74 M Titanium dioxide Lotion 13 ND*** 133.75 -+ 36.08 N Titanium dioxide Lotion 4 ND*** 2.66 + 0.09 O Octyl methoxycinnamate Lotion 2 ND*** 4.77 + 0.8 P Octyl methoxycinnamate Lotion 6 ND*** 36.19 + 11.73 Benzophenone-3 Q Octyl methoxycinnamate Lotion 8 ND*** 18.17 + 15.52 Benzophenone-3 R Octyl dimethyl PABA Lotion 20 ND*** 84.7 + 17.62 Octyl methoxycinnamate Benzophenone-3 Titanium dioxide * In vivo SPF. ** From Eq. 3. *** ND = not determined.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)