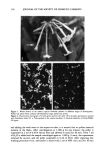
INHIBITION OF ODC INDUCTION 135 15 10 r=0.86 pO.001 0'7 8 ø 0 6 17 50 0 2 0 11 0 4 16 10 0 19 o l•_t20 ø ø9 ø3 o ol •, 26 25 24 21-23 20 15 • 0 O, , , (30 , OO , , 0 , , • • -100 -80 --60 --40 -20 0 20 40 60 80 100 Inhibition of Acute UVR-Induced ODC Activity Figure 2. Correlation between inhibition of UVR-induced ODC activity and delay in onset until 50% incidence (percent of mice with at least one tumor). Point numbers are identified in Table I. inhibiting ODC and yet we observed no effect on tumor latency. Naproxen did lower the tumor yield relative to the vehicle control group. Black and Tigges (25) found that while oral butylated hydroxyanisole (BHA) significantly inhibited UVB-induced epi- dermal ODC, oral BHA had no effect on prevention of UVB-induced skin tumors. More recently, topically applied BHA was shown to have no effect on UVB-induced epidermal ODC activity in mice (27). Three test substances (compound 24, citric acid compound 25, oxothiazolidine car- boxylic acid and compound 26, 4,4'-dipyridyl) increased UVR-induced ODC activity relative to the vehicle control and therefore displayed "negative inhibition." These three compounds had no effect on chronic UVR-induced skin tumor formation. Therefore, while agents that inhibit UVR-induced ODC activity are often photoprotective in the chronic assay, agents that increase UVR-induced ODC activity are not always tumor- promoting. This is consistent with the notion that induction of ODC is a necessary but not sufficient condition of tumor promotion (10). We recognize that the use of two different light sources in the ODC (solar simulator: UVA q- UVB) and tumor (fluorescent suniamps: primarily UVB) studies detracts from the correlation between the two assays. In fact, we have observed subtle differences in the ODC induction profiles by the solar simulator and the suniamps (20). While ODC activity peaks at similar times following exposure to either light source (3 h and 24 h post UV), differences are observed in the ratio of ODC activity at 24 hours vs 3 h post UVR exposure (24-h/3-h ODC activity greater for suniamp induction). However, since exposure of mice to high doses of UVA radiation does not induce ODC nor does UVA
136 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS modulate UVB radiation induction of ODC (20), and since chronic exposure to simu- lated solar radiation induces tumors in hairless mice (1,21), we believe similar results would have been obtained had we used the same light sources for both the acute ODC and the chronic tumor assays. Topically applied sunscreens are known to inhibit UVR-induced ODC (28) and UVR- induced skin tumors (26) in hairless mice. We cannot exclude the possibility that the compounds tested in this study might also inhibit UVR-induced ODC and skin tumors via a sunscreen (UVR absorption) as opposed to a non-sunscreen mechanism of action (e. g., prevention of UVR-induced oxygen radicals). Even the lack of a UV chromophore on many of the compounds tested does not rule out a sunscreen mechanism of action. We are aware of at least two compounds, butylated hydroxytoluene and dihydroxy- acetone, that inherently are not strongly UV-absorbing but, when applied topically, increase the spectral absorption of mouse epidermis (29,30). One approach we have taken to determine the contribution of UV absorbance to the total photoprotective effect is to measure the ability of the test material to inhibit UVR- induced ODC when topically applied to the mice after UVR exposure. In a separate experiment, when two of the iron chelators, dipyridylamine (DPA, compound 1) and furildioxime (FDO, compound 6), were applied immediately after UVR exposure, ODC induction was inhibited by 95% and 21%, respectively (vs 100% and 85%, respec- tively, when applied pre-UVR exposure). The lower level of inhibition observed in the post-UVR experiment could be due to UV absorbance by the test material. An alter- native explanation is that optimum photoprotection is achieved when the iron chelator is present in the skin during UVR exposure when highly reactive oxygen radicals (31) are being generated. In another experiment, we found that both DPA and FDO sig- nificantly inhibited topical 10% benzoyl peroxide induced-ODC activity (100% and 32%, respectively). The observation that DPA and FDO inhibit UVR-induced ODC in a post-UVR treatment protocol and also inhibit chemically-induced ODC suggests that a non-sunscreening mechanism(s) of action plays a significant part in the protection provided by these compounds. We also note that 4,4'-dipyridyl (compound 26) is the non-chelating analog of 2,2'-dipyridyl (compound 19). Both 4,4'- and 2,2'-dipyridyl have similar UV absorbance spectras and extinction coefficients, yet the non-chelating 4,4'-dipyridyl shows no photoprotective efficacy while the chelating 2,2'-dipyridyl displays moderate photoprotection. This strongly suggests that 2,2'-dipyridyl is pho- toprotective, at least in part, via a non-sunscreen mechanism of action. These data can be used to make a general assessment of the relative effectiveness of the molecular classes tested here. Anti-oxidants provide about 50% protection in these assays. Anti-inflammatory agents are approximately equivalent to the anti-oxidants. In general, iron chelators were the most effective agents. Because of the high level of protection observed for iron chelators (24), we have evaluated many more of these than of the other two molecular classes. While chelators provided the highest observed levels of protection, effectiveness covered a wide range, from 0 to 100% protection. The proposed mechanism of protection by chelators is prevention of iron-catalyzed production of oxygen radicals (24). While chelation of iron is a prerequisite for performing this function, not all iron chelators prevent iron-catalyzed production of oxygen radicals (31,32). Given that chelators vary considerably in terms of preventing oxygen radical formation, a wide range of efficacy is expected and observed.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)