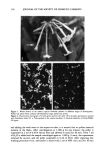
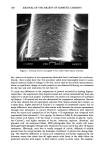
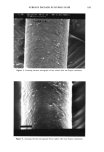
174 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS more susceptible to cuticle loss due to abrasion but also can partially dissolve the sloughed-off protein material during shaking to give lower turbidity readings. Other values presented in the table show the same trend, indicating that a lower turbidity reading does not necessarily mean less cellular material in the solution. The converse is also true. This information, therefore, suggests caution when using turbidity measure- ments to study hair damage. Obviously, quantitatively measuring the amount of protein in the supernatant yields more reliable data. CONCLUSIONS This paper focuses on the development of a simple and sensitive technique to assess surface damage to human hair caused by various damaging treatments such as bleaching and perming. The method involves quantitatively measuring, via a colorimetric proce- dure, the amount of protein lost during shaking of the hair samples in the presence of a simple medium such as water or a surfactant solution. Our studies, based on the above procedure, demonstrate that significant differences in total protein loss can be observed between undamaged hair and hair damaged by various treatments including bleaching and perming. It was also demonstrated that hair with exposed cortex is more susceptible to protein loss by surfactants than hair with an intact cuticle. We were able to show cuticular damage to hair fibers simply after treatment with diperisophthalic acid or a commercially available home permanent or bleaching kit. Results using bleached/ permed hair taken directly from individual heads further complement the validity of this procedure. Previously, diperisophthalic acid-induced surface damage to hair could not be detected using tensile property measurements even after six treatments (wet or dry) (4). However, with this procedure we were able to demonstrate oxidative damage to hair after only three such treatments (results for one treatment were just under the signifi- cance level, p = 0.07). REFERENCES (1) R. Beyak, F. Meyer, and G. Kass, Elasticity and tensile properties of human hair. I. Single fiber test method. J. Soc. Cosine. Chem., 20, 615-626 (1969). (2) R. Beyak, G. S. Kass, and C. F. Meyer, Elasticity and tensile properties of human hair. II. Light radiation effects, J. Soc. Cosmet. Chem., 22, 667-678 (1971). (3) E. Tolgyesi, Weathering of hair, Cosmet. Toiletr. 98, 29-33 (1983). (4) C. R. Robbins and R. J. Crawford, Cuticle damage and the tensile properties of human hair, J. Soc. Cosmet. Chem. 42, 59-67 (1991). (5) J. A. Swift and B. Bews, The chemistry of human hair cuticle--I: A new method for the physical isolation of cuticle, J. Soc. Cosmet. Chem. 25, 13-22 (1974). (6) L. M. Dowling, L. N. Jones, and I. H. Leaver, TEM and X-ray photoelectron spectroscopy studies of wool fibers after cuticle removal, Tex. Res. J., 58, 640-645 (1988). (7) O. H. Lowry, N.J. Rosenbrough, A. L. Farr, and R. J. Rendall, Protein measurements with Folin phenol reagent, J. Biol. Chem., 193, 265-275 (1951). (8) D. H. Speckman, W. H. Stein, and S. Moore, Automatic recording apparatus for use in the chro- matography of amino acids, Anal. Chem., 30, 1190-1206 (1958). (9) C. R. Robbins, "Chemical composition of the Different Morphological Components," in Chemical and Physical Behavior of Human Hair (Springer-Verlag, New York, 1988), pp. 50-53. (10) L. J. Wolfram and M. K. Lindemann, Some observations on hair cuticle, J. Soc. Cosmet. Chem., 22, 839-850 (1971).
SURFACE DAMAGE IN HUMAN HAIR 175 (11) J. A. Swift and A. C. Brown, The critical determination of fine changes in the surface architecture of human hair due to cosmetic treatments. J. Soc. Cosmet. Chem., 23, 695-702 (1972). (12) C. R. Robbins, Chemical aspects of bleaching human hair, J. Soc. Cosmet. Chem., 22, 339-347 (1971). (13) M. Oku, H. Nishimura, and H. Kanehisa, Dissolution of proteins from hair (I). The analysis of dissolved proteins from bleached hair, J. Soc. Cosmet. Chem. of Japan, 21, 198-203 (1987). (14) M. Oku, H. Nishimura, and H. Kanehisa, Dissolution of proteins from hair. (II). The analysis of proteins dissolved into permanent waving agent and the evaluation of hair damage, J. Soc. Cosmet. Chem. of Japan, 21, 204-209 (1987). (15) M. Harris and A. Smith, Oxidation of wool: Alkali-solubility test for determining the extent of oxidation, J. Res. Nat. Bur. Stands., 17, 577 (1936). (16) M. Pelczar and R. Reid, "Determination of Cell Density by Turbidimetric Determination," in Microbiology (McGraw-Hill, New York, 1972), pp. 134-135. (17) E. Layne, "Spectrophotometric and Turbidimetric Methods for Measuring Proteins," in Methods in Enzymology, S. Colowick and N. Kaplan, Eds. (Academic Press, New York, 1957), vol. III, pp. 447-454.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)