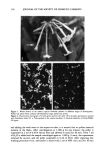
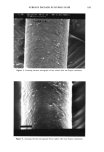
SPF EVALUATION 149 where: A = absorbance. a = absorption. b = path length. c = concentration. A derived form of Beer-Lambert's law (21) expressing the theoretical expression of SPF for a single wavelength (SPFTh) is: SPFTh = 10 •5^ (4) Even when the erythemal action spectrum of human skin is accounted for in the spectrophotometric methods above, the in vitro SPF estimates are much larger than the corresponding in vivo values, suggesting that there are other factors affecting the results besides the simple relationship of Beer-Lambert's law (21). For example, a sunscreen product with an SPF of 4, when measured at a dilute solution (0.1 g/L), would typically have an absorption at 308 nm of 0.4, which would result in a theoretical SPF of 1 million according to Eq. 4 above (21). The characteristics of sunscreen film on skin deviate from a dilute solution of sunscreen upon which the Beer-Lambert relationship is built. The concentration of active sun- screening agents in the film left on the skin after application is high. For example, with 7% w/v Padimate O in a sunscreen formulation, the concentration of the active ingre- dient would be approximately 0.25 m (assuming no evaporation of sunscreen vehicle after application). Beer-Lambert's law maintains a linear relationship only at concen- trations less than 0.01 m. In addition to the high concentration of active present, the film distribution on the skin surface tends also to be uneven (22). A sunscreen vehicle may cause the stratum corneum to hydrate and become more transparent, thereby increasing the transmission of light to the deeper layers of the skin and enhancing erythema. By changing the absorption qualities of the skin, the erythe- mal action spectrum may also change, thus altering the erythemal response. In contrast, the sunscreen vehicle may also increase the reflectance of light from the skin surface by incorporating solid reflectants, pigments, or the skin's refractive index. Components in the sunscreen vehicle itself may interact with the active sunscreen ingredients to cause shifts in the absorption spectrum of the active ingredients (23). Biological reasons for the deviation from Beer-Lambert's law include the erythemal action spectrum and the surface of the skin. An individual's response to UV light is unique and causes differences in the erythemal action spectrum, which in turn influences the SPF determination. Variations in the artificial light sources used to determine SPF can also cause differences in the SPF value (14). Another factor causing a deviation from the application of Beer-Lambert's law in computing SPF relates to the nature of light. The law refers only to monochromatic light, and the situation where polychromatic light is present is more complicated. This is due to the synergy of radiation by multiple wavelengths on the biological response of the skin that is not present under the con- ditions of monochromatic light (24). In addition, the biological response of skin to polychromatic light is not yet fully defined. Finally, the uneven surface topography of the skin (25) makes forming a uniform film of sunscreen a difficult task (20).
150 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS CONCLUSIONS Based on this research, a modified version of the experimental setup may yield better results. Since the sensitivity of the detector used in this study was insufficiently sensitive to distinguish the difference between or among several high-SPF sunscreen products, the use of a more sensitive detector should be investigated. A scanning spectroradiometer used in conjunction with the algorithms for global irradiance and the erythemal action spectrum may help to mitigate the issue of detector sensitivity. Another area warranting investigation relates to the development of alternative substrates for in vitro SPF testing. Custom ground quartz glass plates may be a feasible substrate for in vitro SPF determi- nations because they would eliminate the effect of absorption of sunscreen into the substrate while at the same time providing a "bumpy" surface to mimic the surface topography of the skin. Quartz glass would also provide the necessary UV-transparent qualities so that determinations with a solar simulator as a source of radiation and a radiometer or spectroradiometer could be used as instrumentation. In addition, these plates could be washed and reused. SUMMARY The use of surgical adhesive tapes as substrates for determining SPF in vitro has limi- tations. Transpore © is perforated, and in vitro estimates of SPFs are potentially depen- dent on the sunscreen application technique. The non-perforated surface of Blenderm © tape circumvented the perforation problem but did not significantly increase the cor- relation between the in vitro and in vivo SPFs. However, the fact that the chemical sunscreens absorb into both the tapes is also a problem. Oil- and alcohol-based products give especially erratic results, probably due to vehicle interactions with the tapes. In addition to the substrate limitations, differentiation among sunscreens with high SPFs depends on the sensitivity of the radiometer used to measure UV light transmission. In general, the method used in this study was dependent on the sensitivity of the detector and did not provide a sufficiently precise means of estimating in vitro the SPF of sunscreen formulations. REFERENCES (1) F. Wurst, Th. Prey, J. Washuttl, and F. Greiter, Studies on the adhesive power of several UV filter substances on sheep's wool before and after UV radiation, Arztl. Kosmetologie, 8, 144-154 (1978). (2) F. Greiter, P. Bilek, S. Doskoczil, J. Washuttl, and F. Wurst, Methods for water resistance testing of sun protection products, Int..J. Cosmet. Sci., 1, 147-157 (1979). (3) R. M. Sayre, P. Poh Agin, G. LeVee, and E. Marlowe, A comparison of in vivo and in vitro testing of sunscreening formulations, Photochem. Photobid., 29, 559-566 (1979). (4) R. M. Sayre, P. Poh Agin, D. L. Desrochers, and E. Marlowe, Sunscreen testing methods: In vitro predictions of effectiveness,.J. Soc. Cosmet. Chem., 31, 133-143 (1980). (5) C. Cole and R. Van Fossen, Rapid in vitro evaluation of sunscreens: SPF and PFA, Photochem. ?hotobiol., 47S, 73S (1988). (6) C. Cole and R. Van Fossen, "In Vitro Models for UVB and UVA Photoprotection," in Sunscreens: Development, Evaluation, and Regulatory Aspects, N. J. Lowe and N. A. Shaath, Eds. (Marcel Dekker, New York, 1990), pp. 395-404. (7) J. Ferguson, M. W. Brown, A. W. Hubbard, and M. I. Shaw, Determination of sun protection factors. Correlation between in vivo human studies and an in vitro skin cast method, Int..J. Cosmet. Sd., 10, 117-129 (1988).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)