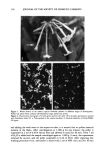
INHIBITION OF ODC INDUCTION 131 Table I Compounds Tested for Inhibition of Acute UVR-Induced ODC Activity and Chronic UVR-Induced Skin Tumors Point number in Figs. 1 and 2 Compound Vehicle Chelators 1 5 % 2,2'-Dipyridylamine PG (propylene glycol): EtOH:H20 (1:2:1) 3 5% 1-Hydroxy-4-methyl-6-(2,4,4-trimethyl- PG:EtOH:H20 (1:2:1) pentyl)-2(1H)-pyridone 4 5 % Di-2-pyridylketone EtOH 5 5 % Diethyldithiocarbamic acid PG:EtOH:H20 (1:2:1) 6 5 % 2-Furildioxime PG:EtOH:H20 (1:2:1) 7 5% 2,4,6-Tri(2-pyridyl)- 1,3,5-triazine EtOH 8 5 % 1-pyrrolidinecarbodithioc acid PG:EtOH:H20 (1.' 1:2) 11 5 % 1, l'-Carbonyldiimidazole EtOH 13 5 % 2,3-Bis(2-pyridyl)-pyrazine EtOH 16 1% 2,2'-Dipyridylamine PG:EtOH:H20 ( 1:2:1) 17 2% 3-(4-phenyl-2-pyridyl)-5-phenyl- EtOH:DMIS (isosorbide 1,2,4-triazine dimethyl ether) (1:1) 19 5 % 2,2'-dipyridyl EtOH 21 5 % 2,3-Di-2-pyridyl-2,3-butanediol EtOH 22 2% 3-hydroxypicolinamide Acetone 23 5 % Benzoin alpha-oxime Acetone:Isopropanol (1:1) 24 5% Citric acid PG:H20 (1:3) 26 5% 4,4'dipyridyl (chelator analog) EtOH Anti-inflammatory agents 2 2% Flufenamic acid PG:EtOH:H20 (1:2:1) 9 5 % Sorbohydroxamic acid PG: EtOH: H20 ( 1: 2:1) 10 0.5% Hydrocortisone EtOH 15 1% Naproxen PG: EtOH: H20 ( 1:2:1) 18 1% Ibuprofen PG:EtOH:H20 (1:2:1) Anti-oxidants 12 5% Tocopherol sorbate Isopropanol 14 5% Alpha-tocopherol EtOH 20 5% Commercial solution of anti-oxidant H20 mixture: lactoferrin + 2-thioxanthine + 8-hydroxyxanthine 25 5% L-2-oxothiazolidine-4-carboxylic acid PG:EtOH:H20 (1:2:1) The procedure for acute UVR exposure of the dorsal skin of mice for induction of ODC has been described previously (20). Mice were individually UVR-exposed with a Kratos solar simulator (Ramsey, NJ) equipped with a 1000-W xenon arc lamp and dichroic filter (UVB q- UVA, 290-400 nm). Mice were placed within the 7-cm diameter circular beam of uniform irradiance (-+ 10%), with the distance from the dichroic mirror to the dorsal surface of the mouse being 10 cm. The UVB q- UVA irradiance at this distance was 18 mW/cm 2. Mice were exposed once to 1.2 J/cm 2 [2 x MED (minimum erythemal dose)] of simulated solar radiation. Mice were sacrificed by cervical dislocation for assay of epidermal ODC 24 h post-UVR
132 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS exposure. The UVR-exposed dorsal skin (•2 x 3 cm) was removed and immediately placed dermal side down on an ice-cold glass plate. The epidermis was scraped off using a single-edged razor blade until the shiny basement membrane was clearly visible. The epidermis was transferred to a tube containing 0.6 ml cold homogenization buffer [50 mm sodium phosphate (pH 7.0), 1.25 mm EDTA, 2.5 mm dithiothreitol, and 0.1 mm pyridoxal 5'-phosphate] and homogenized on ice with a Tekmar Tissumizer (Cincinnati, OH) at setting 8 for 20 s. The homogenate was then centrifuged at 16,000 x g for 10 min at 4øC to obtain the soluble epidermal fraction. ODC activity in the soluble fraction was determined by measuring the release of 14CO 2 from L-[1-•4C]ornithine hydrochlo- ride (New England Nuclear, Boston, MA). The assay mixture (0. 125 ml) contained 40 mm sodium phosphate (pH 7.0), 1 mm EDTA, 2 mM dithiothreitol, 0.2 mM pyridoxal 5'-phosphate, 0.4 mM L-ornithine, 0.5 •Ci of L-[ 1- •4C]ornithine hydrochloride, and 100 •I epidermal soluble fraction. The reaction was started by the addition of substrate and incubated at 37øC for 30 min in 15- x 18-mm culture tubes equipped with a rubber stopper and center well assemblies (Kontes, Vineland, NJ). The reaction was stopped by injecting 0.25 ml of 2 M citric acid (pH 1.5) through the rubber stopper with a syringe and needle. Reactions were allowed to postincubate for at least 1 h at 37øC to ensure complete absorption of 14CO 2 by the methylbenzethonium hydroxide (0.1 ml 1 M in methanol) contained in the center well. Center wells were then transferred to vials containing 10 ml ScintiVerse II scintillation fluid (Fisher Scientific, Pittsburgh, PA), and radioactivity was measured with either a Beckman LS8100 or Wallac 1410 (Pharmacia) liquid scintillation counter. Assays were carried out in du- plicate for each mouse. Blank assays contained homogenization buffer instead of the epidermal soluble fraction, and these values were subtracted from test samples. Protein in the soluble fraction was quantified using the Bio-Rad protein assay (Bio-Rad, Rich- mond, CA), with bovine serum albumin as protein standard. ODC activity was ex- pressed as nmol CO2/h per mg protein. ODC data were expressed as percent inhibition of induction by a test material relative to the vehicle control: [ODC test material - ODC no UVR control] 1 - [ ODC vehicle - ODC no UVR control ] X 100% Typical basal (no UVR control) and UVR-exposed control ODC specific activities were 28 and 662 pmole CO2/h per mg protein, respectively. Treatments, UVR exposure, and sacrifice were carried out the same time each day to help prevent circadian-related effects on ODC activity. CHRONIC TOPICAL TREATMENTS, UVR EXPOSURE, AND QUANTIFICATION OF TUMORS For chronic topical treatment of mice, test substances were applied to the dorsal skin of the mice (n = 8 to 15 per group neck to tail area) in 0.1 ml of test solution (w/v % in the same simple vehicle used in acute studies) 2 h prior to each irradiation. The method of delivery of the test solution to the skin was as described above for acute treatment. The procedure for chronic exposure of the dorsal skin of mice with UVB radiation (290-320 nm) to induce tumors has been described previously (1,2). Mice were indi-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)