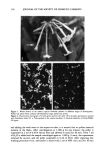
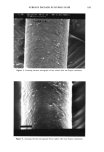
164 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS from the cortex (5,6). We have extended this work to show that the damaged hair surface is more susceptible to abrasion/erosion and that quantitative measurements on the amount of protein removed from hair during abrasion/erosion can serve as a sensitive method to study hair damage. MATERIALS AND METHODS Hair fibers used in this study were purchased from DeMeo Brothers or from private individuals. All hair samples used in various experiments were washed with a mild (5 %) solution of sodium laureth-2 sulfate (SLES) in warm tap water (105øF), rinsed exten- sively with deionized water, and dried with a hair drier. Suprox (consisting of 15.4% diperisophthalic acid encapsulated in hydrated magnesium sulfate and sodium sulfate) was a product of PPG Industries Inc. Reagents for protein estimation were obtained from Sigma Chemical Co., St. Louis, MO. All other reagents used were of analytical grade. TREATMENT OF HAIR WITH SUPROX Ten-inch-long dark brown hair tresses, each weighing 1 g, were treated in triplicate with 2.5 g of Suprox solution for 10 minutes per treatment. Suprox solution was prepared fresh immediately prior to use by mixing 1.3 g of Suprox with 8.1 g of SLS solution (3.85% in water) and 0.6 g of sodium carbonate. Hair tresses were rinsed extensively with warm (105øF) tap water after each treatment. Suprox-treated hairs, dried at room temperature, were cut into small pieces (--•/4 inch length) and stored in glass vials for further experimental use. Finally, Suprox-damaged hair fibers selected at random were also examined under the Amray 1645 scanning electron microscope to visually observe the damage. BLEACHING AND PERMING OF HAIR SAMPLES To study hair damage due to cosmetic treatments, namely, bleaching and perming, 10-inch-long dark brown hair tresses, each weighing 3 g, were permed and/or bleached using commercially available products and following manufacturers' instructions. The bleaching kit contained hydrogen peroxide (Miss Clairol #26S, Winter Wheat by Clairol Inc.), and the permanent wave kit (Ogilvie Home Permanent by Ogilvie Prod- ucts, Inc.) contained ammonium thioglycolate (reducing agent) and hydrogen peroxide (neutralizer) as the active ingredients. The products were applied in sufficient amount to fully saturate the tresses. Following the bleach treatment (90 minutes), the dark brown hair turned light brown in color, with red tones. In addition, damaged and undamaged hair samples, about 2-3 inches in length, were also taken directly from people's heads, cut from the distal end. Damaged hair samples were supplied by indi- viduals who had recently either bleached or permed their hair, whereas the undamaged hair had had no chemical treatment by the donor other than normal shampooing/ conditioning.
SURFACE DAMAGE IN HUMAN HAIR 165 EVALUATION OF HAIR DAMAGE To evaluate hair for surface damage, each hair sample was cut into small pieces • 1/4" in length. 150-300 mg of hair were suspended in 10-25 ml of distilled water in a narrow-mouthed 50-ml Erlenmeyer flask. Each flask was corked and shaken on the Lab Line R multi-wrist action shaker (Lab Line Instruments Inc., Melrose Park, Ill) at a speed arbitrarily set at --75% of the top setting on the (unmarked) scale. Hair samples were shaken for a period of four hours for most of the studies. This was based on our observations, particularly on chemically damaged hair, suggesting that the said time period not only provided an adequate amount of protein concentration in the solution for analysis but also that a major portion (roughly 50% or greater) of the total cuticular material lost during shaking in any given 24-hour period was recovered within the first four hours of shaking. At the end of the shaking period, the water solutions were visibly turbid, containing a very fine suspension of cuticular material, while the hair tended to mat together at the bottom of the flask. The suspended/solubilized protein was piperred off, solubilized under alkaline conditions (see below), and analyzed for protein concen- tration. ESTIMATION OF HAIR PROTEIN Protein concentration in a given sample containing hair protein recovered during shak- ing was determined by the Lowry Method (7), one of the most widely used techniques for protein measurement in biological samples, modified to suit our needs. To solubilize the protein fragments, each sample was well shaken by hand and 0.5 ml of the turbid solution was pipetted directly from the flask and added to a 16 x 125-mm test tube containing 0.5 ml of 1 N NaOH (for samples suspected to contain protein in very low concentration, 1 ml of the sample was used and mixed with 0.1 ml of 5 N NaOH). The contents of the tube were mixed well and allowed to sit at room temperature for 30 minutes to solubilize the suspended protein. At the end of the incubation period, 1 ml of Cu-carbonate solution was added. Cu-carbonate solution was prepared fresh every day by mixing 1 ml each of CuSO4 solution (1% w/v) and potassium tartrate solution (2% w/v) with 20 ml of Na2CO 3 solution (10% w/v). The tubes containing Cu-carbonate- treated alkaline protein solution were incubated at room temperature for 15 minutes. At the end of the incubation period, 3 ml of Folin-phenol solution, prepared by diluting 5.0 ml of 2N Folin-phenol reagent (Sigma Co.) with 50 ml of distilled water, was added and mixed well immediately. The samples were further incubated at room temperature for 40 minutes. At the end of the incubation period, the absorbance of each sample was determined in a spectrophotometer at a wavelength of 750 nm. Protein concentration in each sample was determined from a standard curve (Protein Conc. •g/ml = Slope x Absorbance - Intercept on Y axis this value was then used to calculate the total protein recovered as mg/g hair) prepared separately using crystalline bovine serum albumin (Sigma Co.) as a standard and assayed under conditions identical to the test samples. To check the validity of the method, samples analyzed in triplicate, as well as repeat analysis of a given sample, provided essentially the same results. Using this procedure, we were able to determine as little as 5 micrograms of hair protein per ml in a given solution. The sensitivity of the method can, however, be increased with reasonable precision, if needed, to as little as 1 •g of protein by certain modifications such as
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)