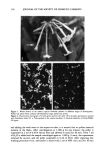
172 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Table V Effect of Various Surfactants on Protein Loss From Hair Surfactant (5% AI) Total protein loss Statistics (mg/g hair s) (ANOVA) Sodium lauryl sulfate (SLS) Ammonium lauryl sulfate (ALS) Water (control) Ammonium lauryl ether sulfate (ALES) Sodium lauryl ether sulfate (SLES) Sodium deceth 3 sulfate (SDES) 10.26 -+ .50 * 9.36 -+ . 19 ** 8.42 +- .29 • 8.12 + .80 7.58 + .23 6.69 +- 1.0 • Each value represents a cumulative average of multiple (6-15) samples and/or experiments. 250 mg of hair shaken in 25 ml of surfactant solution (or distilled water for the control) for 24 hours. * Significantly different from all other data except ALS of this experiment at p = 0.05 level. ** Significantly different from all other data except SLS, water, and ALES at p = 0.05 level. Bar indicates no significant difference at p = 0.05 level. more susceptible to SLS-induced protein loss as compared to undamaged hair. In par- ticular, hair with exposed cortex is more susceptible to abrasion/erosion in the presence of SLS than hair containing intact cuticle. Table VI summarizes the results of Suprox X20-treated hair vs control hair with intact cuticle when shaken in SLS and water. Statistical analysis (3-factor analysis of variance or 3-way ANOVA at p = 0.0001) showed that (i) the amount of protein solubilized in the presence of SLS is significantly greater as compared to water, (ii) Suprox-damaged hair is more susceptible to protein loss than undamaged hair, (iii) Supfox-damaged hair lost more protein in the presence of SLS than water, as compared to undamaged hair shaken under similar conditions (this conclusion is further supported by the fact that the ratio between the amount of protein solubilized by SLS vs water increased from 1.26 for the undamaged hair to 1.72 for Suprox-damaged hair at the end of the two-hour shaking period, suggesting that the damaged hair is more susceptible to loss of protein than the undamaged hair in the presence of a surfactant), and (iv) the amount of protein lost from damaged hair increased significantly over time as compared to undamaged hair under similar experimental conditions, suggesting that damaged hair is more susceptible to protein loss with time. Table VI Effect of Surfactant on Protein Loss From Supfox X 20-Damaged and Undamaged Hair Total protein loss (mg/g hair) Ratio Time (hr) Hair type Water SLS (SLS vs water) 2 Undamaged control hair 2.67 -+ .05 3.37 + . 12 1.26 Supfox-damaged 5.44 + . 19 9.33 + .31 1.72 4 Undamaged control hair 3.04* +- .07 3.96* -+ . 19 1.29 Supfox-damaged 7.06* -+ .45 10.30' -+ .42 1.46 6 Undamaged control hair 4.13' + .01 4.72* + . 19 1.14 Suprox-damaged 9.75* +- .25 12.24' -+ .66 1.26 * Statistical analysis (3-factor analysis of variance or 3-way ANOVA) shows that the observed differences are significant (p = 0.0001) for the detergent, damage, and time. Undamaged hair used in the above experiment is control hair before/without any Supfox treatment, whereas Supfox-damaged hair represents hair from the same bundle following 20 treatments with Supfox.
SURFACE DAMAGE IN HUMAN HAIR 173 These results show that the cuticle is more resistant than the cortex to the degradative effects of abrasion/erosion, especially in the presence of SLS, and are consistent with the effect shown earlier where shaking Suprox x20-treated hair in water produced more protein than hair with intact or partly degraded cuticle. Such an effect is also consistent with the known greater resistance of the cuticle to alkali solubility (15). TURBIDITY VS PROTEIN LOSS MEASUREMENTS AS AN INDEX OF HAIR DAMAGE Turbidity, which measures the degree of opacity of a suspension, is often used to quantify various biological specimens such as bacteria and proteins (16, 17). Shaking hair in a given medium such as water or a surfactant solution produces a turbid supernatant containing hair protein, and one might expect a direct relationship between turbidity and the amount of protein present in the supernatant. However, our studies indicate that turbidity measurements on supernatants containing hair protein removed during shaking of hair samples does not always provide a true indication of hair damage. Turbidity is a function of particle size which, depending upon the severity of damage and the solvent medium, may vary from sample to sample. Moreover, some surfactants may partially dissolve the turbid matter obtained during shaking of hair to lower the turbidity values. To investigate this aspect, undamaged and damaged hair samples were shaken in two different media, namely, water and SLS, followed by both turbidity and protein concentration measurements for each solution at different time intervals over 24 hours of shaking. Experimental results for turbidity, expressed as optical density, vs protein concentration in a given solution are shown in Table VII. Higher optical density readings imply higher turbidity. As seen in the table, optical density of 0. 123 (un- damaged control hair shaken in water for two hours) corresponds to a protein concen- tration of 40 •g/ml, whereas the same hair shaken in SLS, although it shows a lower O.D. reading (0.091), was found to have 51 Ixg protein/ml this corresponds to 25% more protein in the solution, with an O.D. value 27% lower as compared to its counterpart in water. The data suggest that a surfactant such as SLS not only makes hair Table VII Optical Density vs Protein Concentration--Effect of Surfactant and Damage on Hair Protein concentration Optical density of supernatant solution* (•g/cc) Time interval Shaking Undamaged Suprox- Undamaged Suprox- (hr) medium (control) damaged (control) damaged 2 Water 0.123 -- 40 82 2 SLS 0.091 0.019 51 140 4 Water 0.171 0.033 46 106 4 SLS 0.133 0.033 60 155 6 Water 0.191 0.053 64 146 6 SLS 0.155 0.030 71 184 24 Water 0. 340 0. 129 95 298 24 SLS 0. 347 0.054 137 357 * 300 mg of hair shaken in 20 ml of water or SLS on a wrist action shaker. Turbidity was measured as % transmission (T) for each sample in the Spectronic 20 spectrophotometer at 600 nm, found to be optimum for turbidity readings. Values were converted to optical density (OD or Absorbance = - log T). The higher the OD, the more turbid the solution.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)