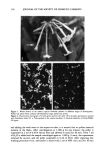
SPF EVALUATION 151 (8) M. Stockdale, A novel assessment of sunscreen product efficacy against UVA, Int. J. Cosmet. Sci., 9, 85-98 (1987). (9) B. L. Diffey and J. Robson, A new substrate to measure sunscreen protection factors throughout the ultraviolet spectrum. J. Soc. Cosmet. Chem. 40, 127-133 (1989). (10) Solar Light Co. Inc., Products description and application. (11) B. L. Diffey, Personal communication (1992). (12) J. DeLuisi, J. Wendell, and F. Kreiner, An examination of the spectral response characteristics of seven Robertson-Berger meters after long-term field use, Photochem. Photobiol., 56, ! 15-122 (!992). (!3) R. M. Sayre, E. Marlowe, L. Desrochers, and F. Urbach, The correlation of indoor solar simulator testing with natural sunlight, Proceedings of the IV Annual Meeting of the American Society for Photobiology, 1976, p. 108. (!4) R. M. Sayre, C. Cole, W. Billhimer, J. Stanfield, and R. D. Ley, Spectral comparison of solar simulators and sunlight, Photodermatol. Photoimmunol. Photomed., 7, 159-165 (!990). (15) R. M. Sayre, J. Powell, and L. A. Rheins, Product application technique alters the sun protection factor, Photodermatol. Photoimmunol. Photomed., 8, 222-224 (1991). (16) S. Brown and B. L. Diffey, The effect of applied thickness on sunscreen protection: In vivo and in vitro studies, Photochem. Photobiol., 44, 509-513, (1986). (!7) K. H. Kaidbey and A.M. Kligman, Laboratory methods for appraising the efficacy of sunscreens,.]. Soc. Cosmet. Chem., 29, 525-536 (1978). (!8) S. I. Kreps, Spectrophotometric evaluation of sunscreens, Proceedings of the Scientific Section of the Toilet Goods Association, 23 (May !955). (19) B. M. Cumpelik, Sunscreens at skin application levels: Direct spectrophotometric evaluation, J. Soc. Cosmet. Chem. 31, 36!-366 (1980). (20) M. Stockdale and D. Roberts, "Multifactorial Influences on Sun Protection Factors," in Sunscreens: Development, Evaluation, and Regulatory Aspects, N.J. Lowe and N. A. Shaath, Eds. (Marcel Dekker, New York, !990), pp. 421-439. (21) M. Stockdale, Sun protection factors, Int. J. Cosmet. Sci. 7, 235-246 (1985). (22) J. J. O'Neill, Effect of film irregularities on sunscreen efficacy,J. Pharm. Sci., 73, 888-891 (1984). (23) L. E. Agrapidis-Paloympis, R. A. Nash, and N. A. Shaath, The effect of solvents on the ultraviolet absorbance of sunscreens. J. Soc. Cosmet. Chem., 38, 209-221 (1987). (24) M. A. Pathak, "Sunscreens: Principles of Photoprotection," in Pharmacology of the Skin, H. Mukhtar, Ed. (CRC Press, Boca Raton, Ann Arbor, London, 1992), pp. 229-248. (25) F. C. Tring and L. M. Murgatroyd, Surface microtopography of normal human skin, Arch. Dermatol., 109, 223-226 (!974). (26) R. M. Sayre, Correlation of in vivo tests, in vitro SPF prediction, a survey of published studies, Cosmet. & Toiletries, 108, 1!1-!!4 (1993).
j. Soc. Cosmet. Chem., 44, 153-162 (May/June 1993) The pollen tube growth test: A new alternative to the Draize eye irritation assay UDO KRISTEN, UDO HOPPE, and WOLFGANG PAPE, Institut fiir Allgemeine Botani K, Universitiit, Ohnhorststrasse 18, D-2000 Hamburg 52, and PGU Research Center, Beiersdorf AG, Unnastrasse 48, 2000 Hamburg 20, Germany. Received January 24, 1992. Synopsis The pollen tube growth test (PTG test), recently developed to detect the cytotoxicity of bioactive chemicals, is compared with the well-established Draize eye irritation assay. The PTG test, based on the growth response of in vitro growing tobacco pollen tubes, is described in detail. Comparison of the corresponding growth response data of both assays, using 22 tensides as test substances, reveals highly significant rank correlations (r = 0.73, p = 0.0018). In both assays, the most toxic substances are among the group of anionic sulfate/sulfonate tensides, whereas the nonionic tensides show moderate effects. The results of the comparison clearly demonstrate that the PTG test is well suited to screen irritation potentials of surfactants and provides reliable results with good reproducibility. Moreover, the PTG test is inexpensive and helps to reduce or even avoid painful animal testing in this application. INTRODUCTION For ethical and economic reasons and because of new animal protection laws, powerful methods must be developed to replace animal experiments. Tissue and cell cultures of animal, human, and plant origin have given good results as alternative systems for toxicological testing of certain classes of substances. However, no alternative method is able to cover the whole range of lethal, sublethal, and chronic toxicity in vertebrates (including man) of all potentially toxic chemical compounds. Of the almost ten million registered substances that could be a toxicological threat to the whole biosphere [ex- trapolated according to Koch (1)], about 0.5% pose a real ecological problem. The potential threat of these 50,000 substances to the environment should be determined by means of suitable toxicological methods while keeping animal experiments to a mini- mum. It is only with a broad range of different alternatives to animal experiments that this can be accomplished. Of the plant organs, tissues and cell types used so far that are suitable for toxicological test methods, the microspores (pollen grains) and their germination product, the pollen tube, have given especially good results. Although germinating pollen grains and 153
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)