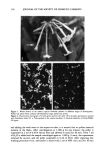
SPF EVALUATION 143 transmission of light (in microvolts) through the tape to the transmission (in microvolts) of attenuated light source intensity. The calculation was performed as follows, with the value of in vitro SPF calculated from Eq. 1 to 3. SPN -- B %TN = 3ALN - B X 100 (1) where: N = the slide position number. %T = percent transmission of light through the tape. SP = slide position average voltage (in microvolts). B = average baseline (in microvolts). AL = attenuated light source average voltage (in microvolts). In vitro SPF = %T untreated surgical tape %T sunscreen-treated surgical tape i•n 1 Z %Ti untreated surgical tape Average in vitro SPF = - n %Ti sunscreen-treated surgical tape i=l (2) (3) where: n = total number of slide sections. ABSORPTION OF SUNSCREEN ACTIVE INGREDIENTS INTO TRANSPORE © TAPE To determine if the sunscreen active ingredients were absorbed into the tape, Transpore © was treated with 2 txl/cm 2 of sunscreen D as previously described. The percent light transmitted through the three slide sections was measured in the manner previously described for in vitro SPF evaluation. Two sunscreen-treated slides were gently washed with a detergent and water solution and light rubbing for 30 seconds to remove the sunscreen from the surface of the tape. The slides were dried for 15 minutes in a 37 -+ 3øC oven, and the percent transmission of light through the tape was determined. The washing, drying, and reading procedure was repeated until a constant percent transmission reading was reached. Each tape was removed from the slide frame and placed in a solution of 50 mL of tetrahydrofuran (THF) for five minutes. The tape was then replaced in the slide frame, and the percent transmission of light through the tape was measured. In addition, the absorbance spectra of the material solubilized in THF was determined spectrophotometrically. DILUTION OF TWO SUNSCREEN FORMULATIONS WITH THEIR VEHICLES ON BLENDERM © TAPE The vehicles for sunscreens for D and G were prepared by omitting the active ingredients from their formulations. The vehicles were used to dilute their respective products, and measurements of the in vitro SPF of the dilutions were made. Dilutions of sunscreen D were made in the following proportions of parts vehicle to parts sunscreen D: 7:1, 3:1, 2:1, 1:1, and 1:2 and dilutions of Sunscreen G were made in the following dilutions: 1:1 and 3:1. The in vitro SPF was obtained on Blenderm © by the method described above
144 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS and Eq. 2. The resulting in vitro SPF values were plotted versus the dilution of the sunscreen. RESULTS AND DISCUSSION The objective of evaluating static SPF on surgical tapes was to evaluate the method of in vitro SPF assessment based on that reported by Diffey and Robson (9) but using different equipment. Unfortunately, Diffey's Transpore © tape technique with the ad- aptation of using the 150-watt solar simulator and 3D radiometer yielded several problems. The in vitro results obtained did not consistently yield SPF values that could be correlated to the label claim SPFs, and the errors associated with the measurements were unacceptably large, particularly for products with low %T (Table I). The in vitro SPF values on Blenderm © and Transpore © from Table I were graphed versus their corresponding in vivo SPFs, and the correlation coefficients (r) were calculated (Figures 2 and 3). In addition to the unacceptable high errors (standard deviations shown as error bars), the correlation coefficients were low (r --- 0.587 on Transpore © tape and r = 0. 397 on Blenderm©). Diffey's correlation coefficient was much higher, r = 0.943 (9). Table II shows the comparison of Diffey's results on Transpore © tape (11) to those obtained in this study and the in vivo SPF values for sunscreens B and D. Diffey's results 45 40 35 30 25 20 15 10 5 0 0 5 10 15 20 25 30 Label Claim SPF ß In Vitro SPF on Transpore © tape - - Best fit line r = 0.587 Figure 2. In vitro SPF on Transpore © tape.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)