154 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS growing pollen tubes react extremely sensitively to changes in their environment, it is relatively easy to maintain a slow growth of pollen tubes for several days in special artificial culture media. Both properties, sensitivity and cultivability, are important requirements for a toxicological test system. Another requirement is reproducibility of a defined response and its measurability. This requirement is also met by in vitro germinating pollen and growing pollen tubes, since both respond with a growth inhibition that can be reproduced well and is easily quantified. The growth inhibition of pollen tubes has already been determined for some time by measuring the length microscopically. Several hundred pollen tubes must be measured to obtain significant results. In the past 20 years, this quantitive microscopic method has been used to study the biological effect of many substances, especially those having an impact on the environment, e.g., SO 2 and fluoride (2), inorganic components of acid rain (3,4), acids and heavy metals (5), and many other compounds from various sub- stance classes (summary in 6). The microscopic method is complicated and time-consuming the pollen tubes growing in a culture medium are usually very bent, making measurements of the length more difficult. The length can be measured faster using a method developed by Iwanami (7) and modified by Martin (8) in which the pollen grains are applied to an agar plate in a straight line. This method gives relatively good estimates of the inhibitory effect of water-soluble compounds but provides no accurate quantitative evaluations. The method developed by us is based on a quantitative photometric measurement of the pollen tube mass produced in vitro. Measuring the length of individual pollen tubes can be avoided by measuring the turbidity of the pollen tube suspension after ultrasonic treatment (9). The efficiency of this photometric method can be increased even further if the concentration of a dye that is bound chemically to the pollen tubes but dissociates when the pH is lowered is measured instead of the turbidity. The Alcian blue dye has been shown to give especially reproducible results in our studies (10). In this paper we present a photometric method for determination of the cytotoxicity of primary water-soluble substances or substances that are soluble in water with the aid of solubilizers, and compare this pollen tube growth test (PTG-test) with the Draize eye irritation assay (! 1) commonly used in cosmetic safety testing. Recently, the PTG test was proven to be useful for cosmetics. In the CTFA (The Cosmetic, Toiletry and Fragrance Association, U.S.A.) Evaluation of Alternative Pro- gram (Phase I and II), the PTG test was among the five best of 30 in vitro alternatives to the Draize primary eye irritation test (12,13). Even less soluble products were successfully handled by the PTG test as evidently shown by the preliminary results of phase II of CTFA's program (Gettings et al., in preparation). The PTG test is also successfully involved in another international validation program, the Multicenter Evaluation of In Vitro Cytotoxicity (MEIC, Sweden), which is directed to evaluate the relevance to human toxicity of more than a hundred in vitro assays (14). Moreover, the PTG test was used to determine the cytotoxicity of airborne organics (15), herbicides (16), and pesticides and other chemicals (17).
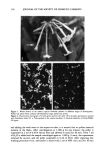
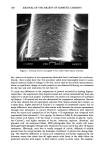
POLLEN TUBE GROWTH TEST 155 MATERIALS AND METHODS POLLEN COLLECTION Tobacco plants of the species, Nicotiana sylvestris (Spegazz & Comes var. SCR) (Figure 1) were cultivated from seeds in a greenhouse and set out to flower in the Botanical Garden of the University of Hamburg. The anthers of flowers that were not yet fully open were harvested and dried for two days at 20øC in a Petri dish. The pollen was obtained by shaking the dried anthers and then freezing them in Eppendorf tubes (1.5 ml) at minus 18øC. The pollen can be maintained in this state for three years with practically no loss of its ability to germinate and can thus be used in toxicity tests at times other than the flowering period. IN VITRO CULTURE OF POLLEN AND INCUBATION WITH TEST SUBSTANCES The culture medium is composed of 10% saccharose (w/v), 0.01% boric acid (w/v), and 3 mM Ca(NO3) 2 in double-distilled water. It is buffered with 10 mM MES (2-[N-mor- pholino]-ethanesulfonic acid) and adjusted to a pH of 5.6 with KOH. The substances tested thus far did not affect the pH. After thawing, the pollen material is homoge- neously suspended in a concentration of 1 mg of dried pollen per 1 ml of culture medium by stirring. While stirring is continued, a 900-1M aliquot of this pollen suspension is pipetted into 50-ml Erlenmeyer screw-topped flasks. The cultures for the test are prepared in duplicate by adding the following to the pollen suspensions in the Erlenmeyer flasks: Control with pure water --Addition of 100 I.tl H20 Control with solubilizer --Addition of 100 I.tl 10% DMSO Zero timer control --Addition of 100 I.tl ethanol Test condition --Addition of 100 I.tl stock solution of test substance. A solubilizer, DMSO (dimethylsulfoxide), must be used if a test substance does not completely dissolve at 25øC. DMSO is used at a final concentration of 1%, which has no influence on pollen germination or pollen tube growth. A zero time control is necessary to eliminate any influence of pollen grains on the photometric values an ethanol concentration of 10% is sufficient to reduce pollen tube growth to zero. For the "test conditions," stock solutions of different concentrations of test substance are used. The cultures are incubated on a shaker for 18 h at 25øC. INCUBATION WITH THE TEST SUBSTANCE, STAINING, AND PHOTOMETRY Usually it is necessary to determine the effective concentration range of a test substance before constructing a dose-response curve. To do this seven different concentrations ranging from 10 mM to 10-5 mM are pretested following the procedure described above. The critical concentration range can be estimated by visually comparing the turbidity of the test suspensions with that of the control, or it can be narrowed down by examination of the suspension with a fluorescence microscope (Figure 2). After this is done, a series of concentrations (each at least in quadruplicate) with small concentration increments (increasing by a factor of 2) is used to narrow down the critical concentration range even further. After incubation for 18 hours, the suspensions in each Erlenmeyer flask are transferred to centrifuge tubes. By rinsing the flasks twice with 4-ml portions of H20
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)